Autor Schiavone Miguel1, Blanco MagalĂ2-31, Ernst Glenda2, Salvado Alejandro2, Manuale Osvaldo1, Nosetto Daniela1 y Borsini Eduardo2-3
1 Arterial Hypertension Unit 2 Center for Respiratory Medicine 3 Sleep and Ventilation Unit. Hospital Británico de la Ciudad de Buenos Aires. Argentina
Correspondencia : Dr. Miguel Schiavone: mjschiavone@gmail.com; Dr. Eduardo Borsini: borsinieduardo@yahoo.com.ar
Abstract
Introduction: Arterial hypertension and obstructive sleep apneas are high prevalence diseases frequently associated. Understanding the hemodynamic profiles would allow treatment administration basing on the changes produced by the combination of both diseases.
Materials and Methods: A prospective, exploratory pilot study was conducted with the purpose of characterizing the hemodynamic patterns of patients referred to the Arterial Hypertension Center who were without pharmacological treatment at the time of consultation. The hemodynamic pattern and thoracic fluid content were evaluated by impedance cardiography. In addition, office and 24-hour ambulatory monitoring of arterial pressure values were recorded; and the suspicion of sleep apnea was objectively assessed by means of home respiratory polygraphy.
Results: 58 patients were included. The diagnosis of sleep apnea was confirmed in 84.5% of the cases (46.5% moderate to severe), and arterial hypertension in 65.2%. The findings of this study showed a progressive decrease in the proportion of the normodynamic pattern and normal thoracic fluid content, in relation to the increase in the severity of the respiratory sleep disorder (p 0.5). Furthermore, office systolic and diastolic pressure showed a progressive increase in relation to the increase in the severity of sleep apnea (p 0.05 and 0.01).
Conclusion: The moderate-severe respiratory sleep disorder was related to an increase in resting blood pressure and a trend that did not reach statistical significance in the findings of altered hemodynamic patterns.
Key words: Arterial hypertension; Sleep apneas; Hemodynamic patterns.
Introduction
Arterial hypertension (AHT) affects 33.5% of the adult population1. It has been shown that this disease is associated with cardiovascular diseases such as heart failure, acute myocardial infarction, renal failure and stroke after years of exposure2.
Sometimes, it is possible to detect the underlying cause that contributes to the increase in arterial pressure (AP). One of those causes is the obstructive sleep apnea and hypopnea syndrome (OSAHS). It has been described that the OSAHS affects approximately 30% of the general adult population3, even though there is a high sub-diagnosis rate4. Thus, the OSAHS represents an emergent sanitary dilemma due to its elevated prevalence and the morbidity and mortality originated by it, which can be attributed to car accidents and the development of cardiovascular complications2-5.
The higher degree of evidence of cardiovascular risk and OSAHS relies upon their association with AHT. Respiratory sleep disorders have also been associated with ischemic cardiopathy, heart arrythmias and pulmonary hypertension. Furthermore, the cause-effect relationship has been strengthened by the identification of possible pathogenic routes, as well as documentation regarding the effects of the treatment on the cardiovascular system2-8.
It has been previously shown that the OSAHS would be an independent risk factor for the development of AHT. Worsnop et al observed a significant association between AHT and an Apnea-Hypopnea Index (AHI) of > 5/hour by means of polysomnography and 24-hour ambulatory monitoring of arterial pressure (AMAP)5. Furthermore, Pedrosa et al showed that 64% of patients diagnosed with secondary AHT had respiratory sleep disorders6.
Patients with AHT show an alteration in hemodynamic factors such as peripheral vasoconstriction, cardiac index or an increase in intravascular volume, even in some occasions associated with the increase in plasma aldosterone levels6-9. Despite this evidence, the hemodynamic characteristics of patients with AHT and OSAHS aren´t known in detail. The results of impedance cardiography studies (ICG) allow us to know the hemodynamic pattern of the patients10-12. This technique could be useful to understand the determinants of the increase in AP and administer the initial anti-hypertensive treatment or adapt the pharmacologic approach.
The main purpose of this study it to characterize the different hemodynamic patterns found in patients diagnosed with AHT and OSAHS, referred to the Arterial Hypertension Center of the Cardiology Service in a community- university hospital.
Materials and Methods
Design: Prospective, exploratory pilot study in patients with presumptive diagnosis of AHT and OSAHS who were without treatment at the time of consultation. The data of the present study represent the preliminary analysis of a bigger study still in progress and have been approved by the Institutional review Board in accordance with the Helsinki rules; CRIHB (Criteria for Review and Approval of Applications) approval N°: #639.
Population Under Study: Between March 2016 and March 2017 we included patients between 18 and 65 years old with presumptive diagnosis of AHT referred for diagnosis and stratification.
Patients with suspicion of OSAHS were defined by: a score of more than 10 points in the Epworth Subjective daytime Sleepiness Scale13 (ESS), a high risk Berlin questionnaire, and/or more than 5 points in any combination of the STOP-BANG14-16 scale, and were considered eligible for the protocol (any of the three options).
We excluded from the study those patients with OSAHS under treatment with CPAP (continuous positive airway pressure) or mandibular advancement devices, and/or with AHT under treatment with drugs. We also excluded patients with morbid obesity (BMI [Body Mass Index] of more than 40 kg/m2) and diseases such as: chronic obstructive pulmonary disease, neuromuscular disease and acute or chronic heart failure.
Baseline Clinical Evaluation: At the beginning of the study we performed an anamnesis of the patient (history and risk factors), anthropometric data (BMI, neck circumference, waist perimeter), office arterial pressure measurement, biochemical blood analysis, electrocardiogram, color doppler echocardiogram, AMAP and ICG. After the Berlin, STOP-BANG and Epworth questionnaires were carried out, patients with high risk of OSAHS underwent a nocturnal respiratory polygraphy (RP).
Arterial Pressure Measurement: The measurement of AP was made according to national guidelines17. It was recorded with an automatic tensiometer (OMRON 7220). After 5 minutes at rest, three measurements were made, separated by 2 minutes, and the average was recorded. Ambulatory Monitoring of Arterial Pressure: 24-hour AMAP was carried out with Spacelabs Ultralite equipment (model 90217, SpaceLabs, Redmond, WA). The AP measurements were set every 15 minutes during the day (8:00 a.m. to 11:00 p.m.) and every 30 minutes during the night (11:00 p.m. to 8:00 a.m.). We defined day-and-night controlled AP when the systolic and diastolic values were less than or equal to 135/85 mmHg and 120/80 mmHg, respectively17. The RP and AMAP were recorded on successive nights.
Those in charge of reading and interpreting the AMAP were cardiologists specialized in AHT. We defined predominance patterns in the AP (systolic/diastolic or systo-diastolic) and the time (day/night or both).
Noninvasive Measurement of Hemodynamic Patterns: The measurement of hemodynamic patterns was made in a noninvasive manner through the ICG. The hemodynamic variables are established basing on the determination of the electric impedance variations of the thorax, induced by changes in the aortic fluid during the cardiac cycle. The measurements were taken bare chested, in the dorsal decubitus position after 30 minutes at rest in the morning, before 09:00 a.m. By means of a transfer equation (Kubicek equation), the systolic volume was calculated. As of this parameter and together with the heart rate and AP, the cardiac output, peripheral resistance and fluid content (TFC) were calculated18.
Respiratory Polygraphy: We used Apnea Link Plus™ polygraphy devices (ResMed. Australia) with five channels and three basic signals, pulse oximetry, nasal cannula flow and thoracic effort (level III devices of the American Academy of Sleep Medicine)19. The reading was made from a distance (Sleep and Ventilation Unit) through sequential manual analysis by pulmonologists with experience in sleep medicine. We considered as valid only those measurements with a total recording time through manual analysis of > 240 minutes (> 4 hours). Apnea was defined as reduction of airflow of > 80% of the basal ≥ 10 seconds (s), and hypopneas were considered as a reduction of airflow of 50% ≥ 10 s associated with desaturations ≥ 3%20. The Apnea/Hypopnea Index (AHI) was calculated as the number of apneas/ hypopneas per hour of valid evaluation of the total recording time (TRT). Patients were classified as normal (AHI < 5/h), mild (AHI ≥ 5 and < 15), moderate (AHI ≥ 15 and < 30) and severe (AHI ≥ 30).
Statistical Analysis: Results were presented as percentages for the categorical variables or as mean and standard deviation for the numerical variables. Normal distribution variables were expressed as mean and standard deviation, and variables without normal distribution are shown as mean and percentiles (25-75%). For the comparison of differences, we used the Mann-Whitney or χ2 or Fisher tests. For the statistical analysis we used Graph Pad Prism-6™ software.
Results
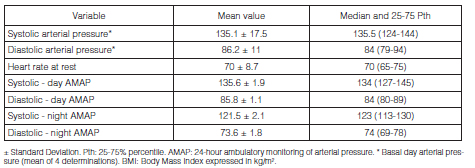
Population: We included in the analysis 58 patients whose demographic characteristics are observed in Table 1. Findings according to the elements present in the STOP-BANG questionnaire are shown in Table 2, and office and AMAP basal measurements of AP can be seen in Table 3. The diagnosis of AHT was confirmed by AMAP in 65.2% of the patients, out of which 13% resulted in isolated nocturnal AHT.
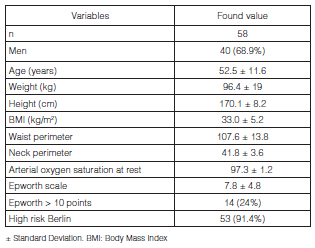
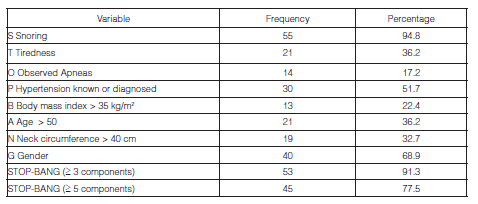
The diagnosis of OSAHS was confirmed in 84.5% of the patients, 46.5% out of which were moderate-to-severe manifestations, categorized as per the AHI. The main reasons for performing the RP were: snoring (94.0%), age > 50 years old (68.3%), daytime tiredness (34.5%), more than 5 points in the STOP-BANG scale (n = 45; 77.5%) and, finally, more than 10 points in the Epworth scale (n = 14; 24%).
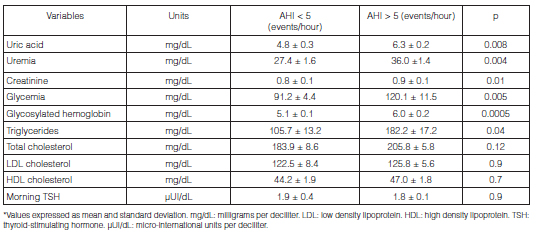
We found a significant increase in the lab values of patients with OSAHS in comparison with patients with an AHI < 5 events/hour regarding glycemia (mg/dL): 120.1 ± 11.5 vs. 91.2 ± 4.4; p < 0.01 (Table 4).
Alteration of the Hemodynamic Pattern in Relation to the Severity of the OSAHS: We observed a progressive decrease that didn’t reach a statistically significant difference in the proportion of patients with normal hemodynamic pattern (normodynamic pattern) in terms of the increase in the OSAHS severity (Figure 1A).
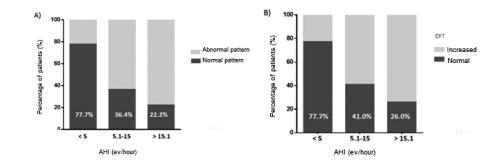
Increase in the Thoracic Fluid Content in Relation to the Severity of the OSAHS: We found a progressive increase in the TFC in terms of the increase in the severity of the OSAHS (Figure 1B). This increase didn’t reach statistically significant differences, either.
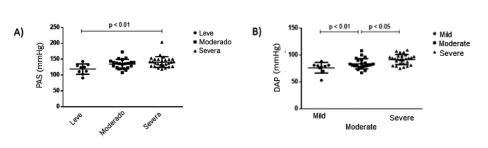
Increase in Office Arterial Pressure in Relation to the Severity of the OSAHS: Both in the systolic arterial pressure (SAP) and the diastolic arterial pressure (DAP) values we observed a progressive increase in relation to the increase in the severity of the respiratory sleep disorder as per the AHI (normal: 130.4 ± 3.2 mmHg and 85.1 ± 2.8 mmHg; mild: 134.2 ± 3.2 mmHg and 85.4 ± 2.3 mmHg and moderate to severe: 135.5 ± 4.1 mmHg and 87.3 ± 2.3 mmHg; p < 0.05 and p < 0.01 respectively) (Figure 2 A and B).
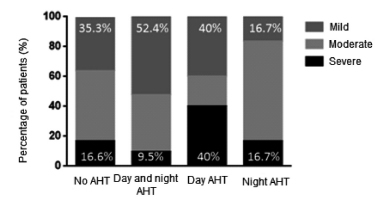
Increase in Arterial Pressure by AMAP in Relation to the Severity of the OSAHS: Although there wasn’t any statistically significant difference, we observed that double the amount of patients with isolated nocturnal AHT suffered moderate to severe sleep apneas, compared to isolated daytime AHT patients (Figure 3).
Discussion
The ICG is a fast, noninvasive method for measuring the hemodynamic profile of hypertensive patients, allowing us to deduce the prevalent physiopathological mechanism and consequently choose the anti-hypertensive treatment. However, there is insufficient information about the hemodynamic pattern present in different phenotypes of hypertensive individuals, not to mention about its relationship with the OSAHS.
The findings of this study carried out in patients with suspicion of AHT without an established treatment showed a trend that suggests a proportional increase relationship between the severity of the OSAHS and an abnormal hemodynamic pattern, not reaching statistical significance.
Our results are relevant because 46.5% of recently diagnosed AHT patients referred to a specialized center present moderate to severe OSAHS and are candidates for treatment with CPAP.
Office SAP and DAP results revealed an increase that is proportional to the severity of the OSAHS; however, mean values of office AP were considered as normotension. These results agree with the existent evidence showing that patients with OSAHS usually have hidden AHT5, 6, 21.
In order to confirm the AP values, we used the AMAP, which showed that 65.2% of the patients were hypertensive, 13% out of which had isolated nocturnal AHT. However, a greater percentage of patients with day AHT in the AMAP showed moderate to severe OSAHS, contributing to alert us about circadian presentation patterns different to the nocturnal pattern of this population.
Pratt-Ubunama has shown the association between the OSAHS and the hyperaldosteronism, producing a higher amount of intravascular fluid due to the reabsorption of sodium and potassium excretion related to aldosterone9. Furthermore, regarding these results, a favorable response has been described in patients with OSAHS towards antialdosteronic agents22.
In accordance with available data, studies carried out in poorly-controlled hypertensive patients show a caudal-rostral fluid shift as a pathogenic route with a significant role in the physiopathology of the OSAHS23. Interaction between liquid shift in patients with AHT and OSAHS is not entirely clear yet23-24. Our data based on the ICG show that TFC is related to the AHI, contributing to understand that it increases proportionally to the severity of the OSAHS. Understanding the hemodynamic profiles would allow for the administration of AHT treatment basing on hemodynamic changes produced by the combination of AHT and OSAHS25.
Our study has multiple limitations. There may be an AHI underestimation between 10 and 15% when using a RP, in comparison with conventional PSG (polysomnography). This represents a limitation of the RP inherent to the absence of neurophysiological signals. The indices that were used (AHI) differ from those of the PSG (AHI or RDI [respiratory disturbance index]), since the AHI results from the quotient between the flow events and the total recording time.
This little study was conducted in a sample of ambulatory adult patients referred to a cardiology service for a specialized evaluation due to suspected diagnosis of AHT. For that reason, the prevalence of OSAHS in that group is higher than that of the general population, though coincident with the data previously reported in our area using RP in a specialized center28. Also, patients were selected due to their elevated office AP values and higher risk of suffering sleep apneas; this fact can explain the high prevalence of both entities and bias the results when comparing the groups. Finally, the low statistical power derived from a small sample requires confirmation by means of studies including a larger amount of patients.
Conclusions
Moderate-severe respiratory sleep disorder was related to an increase in resting blood pressure and a trend that did not reach statistical significance in the findings of altered hemodynamic patterns.
However, new studies with a larger sample size are necessary in order to confirm our findings.
Conflicts of interest: The authors declare they have no conflict of interest with the issue related to this original text.
1. Marín M, Fábregues G, Rodriguez P, y cols. Registro Nacional de Hipertensión Arterial. Conocimiento, tratamiento y control de la hipertensión arterial. Estudio RENATA. Rev Argent Cardiol. 2012; 80: 121-9.
2. Omura M, Yamaguchi K, Kakuta , Nishikawa T. Prospective study on the prevalence of secondary hipertensión among hypertensive patients visiting a general outpatient clinic in Japan.Hypertens Res. 2004; 27: 193-202.
3. Tufik S, Santos-Silva R, Taddei JA, Bittencourt LR. Obstructive sleep apnea syndrome in the Sao Paulo Epidemiologic Sleep Study. Sleep Med. 2010. 11(5): 441-6.
4. Peppard PE, Young T, Palta M, Skatrud J. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med. 2000; 342: 1378-84.
5. Williams AJ, Houston D, Finberg S, et al. Sleep apnea syndrome and essencial hypertension. Am J Cardiol. 1985; 103: 1019- 22.
6. Pedrosa RP, Drager LF, Gonzaga CC, Sousa MG, de Paula LK, Amaro AC, et al. Obstructive sleep apnea: the most common secondary cause of hypertension associated with resistant hypertension. Hypertension. 2011; 58(5): 811-7.
7. Ke X, Guo W, Peng H, et al. Association of aldosterone excess and apnea-hypopnea index in patients with resistant hypertension. Sci Rep. 2017; 22; 7: 45241.
8. De Souza F, Muxfeldt ES, Margallo V, Cortez AF, Cavalcanti AH, Salles GF. Effects of continuous positive airway pressure treatment on aldosterone excretion in patients with obstructive sleep apnoea and resistant hypertension: a randomized controlled trial. J Hypertens. 2017; 35(4): 837-44.
9. Pratt-Ubunama MN, Nishizaka MK, Boedefeld RL, Cofield SS, Harding SM, Calhoun DA. Plasma aldosterone is related to severity of obstructive sleep apnea in subjects with resistant hypertension. Chest. 2007; 131: 453-9.
10. Smith R, Levy P. Ferrario, C, for the Consideration of Noninvasive Hemodynamic Monitoring to Target Reduction of Blood Pressure Levels Study Group: Value of Noninvasive Hemodynamics to Achieve Blood Pressure Control in Hypertensive Subjects. Hypertension. 2006; 47: 771-7.
11. Treister N, Wagner K, Jansen PR. Reproducibility of impedance cardiography parameters in outpatients with clinically stable coronary artery disease. Am J Hypertens. 2005; 18(2): 44S-50S.
12. Taler SJ, Textor SC, Augustine JE. Resistant hypertension: comparing hemodynamic management to specialist care. Hypertension. 2002; 39(5): 982-8.
13. Chiner E, Arriero JM, Signes-Costa J, Marco J, Fuentes I. Validation of the Spanish version of the Epworth Sleepiness Scale in patients with a sleep apnea síndrome. Arch Bronconeumol. 1999; 35: 422-7.
14. Borsini E, Ernst G, Salvado A, et al. Utility of the STOP-BANG components to identify sleep apnea using home respiratory polygraphy. Sleep Breath. 2015; 19(4): 1327-33.
15. Chung F, Yegneswaran B, Liao P, et al. Validation of the Berlin questionnaire and American Society of Anesthesiologists checklist as screening tools for obstructive sleep apnea in surgical patients. Anesthesiology. 2008; 108(5): 822-30.
16. Chung F, Yegneswaran B, Liao P, et al. STOP questionnaire: a tool to screen patients for obstructive sleep apnea. Anesthesiology. 2008; 108(5): 812-21.
17. Consensus on Arterial Hypertension. Argentine Board of Arterial Hypertension “Dr. Eduardo Braun Menéndez”. Argentine Society of Cardiology. Rev Argent Cardiol. 2013; 81: 1-72.
18. Ferrario CM, Basile J, Bestermann W, et al. The role of noninvasive hemodynamic monitoring in the evaluation and treatment of hypertension. Ther Adv Cardiovasc Dis. 2007; 1(2): 113-8.
19. Clinical Guidelines for the Use of Unattended Portable Monitors in the Diagnosis of Obstructive Sleep Apnea in Adult Patients. Portable Monitoring Task Force of the American Academy of Sleep Medicine Journal of Clinical Sleep Medicine. 2007; 3(7): 737-47.
20. Berry RB, Budhiraja R, Gottlieb DJ, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med. 2012; 8(5): 597-619.
21. Kario K. Obstructive sleep apnea syndrome and hypertension: ambulatory blood pressure. Hypertension Research. 2009; 32: 428–432.
22. Gaddam K, Pimenta E, Thomas SJ, et al. Spironolactone reduces severity of obstructive sleep apnea in patients with resistant hypertension: a preliminary report. J Hum Hypertens. 2010; 24: 532-7.
23. Friedman O, Bradley TD, Chan CT, Parkes R, Logan AG. Relationship between overnight rostral fluid shift and obstructive sleep apnea in drug-resistant hypertension. Hypertension. 2010; 56: 1077–1082.
24. Dempsey JA, Veasey SC, Morgan BJ, O’Donnell CP. Pathophysiology of sleep apnea. Physiol Rev. 2010; 90: 47–112.
25. Van De Water JM, Miller TW, Vogel RL, Mount BE, Dalton ML. Impedance cardiography: the next vital sign technology? Chest. 2003; 123(6): 2028-33.
26. Skomro RP, Gjevre J, Reid J, et al. Outcomes of home based diagnosis and treatment of obstructive sleep apnea. Chest. 2010; 138: 257-63.
27. Kuna ST, Gurubhagavatula I, Maislin G, et al. Non inferiority of functional outcome in ambulatory management of obstructive sleep apnea. American Journal of Respiratory and Critical Care Medicine. 2011; 183: 1238-44.
28. Borsini E, Blanco M, Bosio M, Schrappe M, Ernst G, Nosetto D, et al. Prevalence of sleep apnea and cardiovascular risk factors in patients with hypertension in a day hospital model. Clin Exp Hypertens. 2017; 5: 1-7.