Autor : Dell’Era Silvina1, Roux Nicolás1, Gimeno-Santos Elena2, Terrasa Sergio1 Contributors: Castellano Barneche MarĂa Florencia1, Balestrieri MarĂa Carolina1, Gracia Guadalupe1, Bykhovsky Ilona1, Midley Alejandro1
1 Hospital Italiano de Buenos Aires, Argentina 2 Hospital ClĂnic, Barcelona, España
Correspondencia : Silvina Dell’Era. silvina.dellera@hospitalitaliano.org.ar
Abstract
Introduction: The effects of high flow nasal cannula (HFNC) such as reducing of work of breathing could improve exercise tolerance in stable chronic obstructive pulmonary disease (COPD) patients, however there is a great lack of evidence supporting its use.
Aims: To compare the maximum speed (MS) reached on an Incremental Exercise Test (IET) and Limit Time (Tlim) on a Constant Work Rate Exercise Test (CWRET) using High Flow Nasal Cannula (HFNC) vs Venturi Mask (VM).
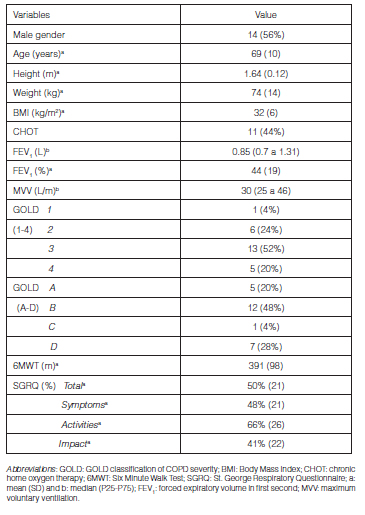
Methods Subjects: 28 COPD patients (56% male, age mean 69 yr and FEV1 44% predicted) who require oxygen during exercise.
Design: randomized crossover trial.
Variables: MS, Tlim, symptoms, oxygen saturation (SpO2), heart rate, respiratory rate, blood pressure and comfort.
Procedure: Each patient performed two IET and two CWRET, one time using HFNC and the other with VM.
Statistic analysis: we carried out a hypothesis contrast of paired data comparing MS, Tlim and physiological variables. p < 0.05 was considered statistically significant.
Results: HFNC vs MV increased MS (5,9 vs 5,7 km/h; p = 0.0002) and Tlim (450 vs 315 sec; p = 0.004), with higher SpO2 and less dyspnea (p < 0.05) at both isowork and isotime, with no statistically significant differences on confort.
Conclusions: HFNC improved exercise capacity in COPD patients increasing MS during IET and Tlim on CWRET with higher SpO2 and reduced dyspnea in comparison to VM. HFNC and VM were equally comfortable.
Key words: High flow nasal cannula; Oxygen inhalation therapies; Exercise training; Rehabilitation; COPD.
Introduction
Patients with chronic respiratory diseases such as chronic obstructive pulmonary disease (COPD), interstitial pulmonary diseases and cystic fibrosis present dyspnea, fatigue and limitation on the activities of daily living (ADL). A decreased exercise capacity, one of the most important systemic consequences, is related to multiple factors such as airflow obstruction, dynamic hyperinflation, alteration of gas exchange and peripheral muscle failure1, 2.
Pulmonary rehabilitation (PR) is one of the main pillars of the treatment of patients with chronic respiratory diseases. Multiple studies showed benefits in the improvement of exercise capacity, muscle strength, symptoms and quality of life3-6. Although the PR programs may vary in duration and components, they must include training aerobic capacity to improve peripheral muscle failure and the symptoms through the increase in muscle capillarization and oxidative enzymes, and reducing the energy and ventilatory cost during the ADL. There is evidence that higher training loads, mainly intensity and duration7, produce greater aerobic training results. In order to plan an optimum, personalized aerobic training load and the assessment of treatment results, we evaluate the exercise capacity of every patient by means of Incremental Exercise Tests (IETs) and Constant Work Rate Exercise Tests (CWRETs), carried out frequently in treadmills or cycle ergometers8.
Several studies documented the benefits of supplemental oxygen during exercise in patients with COPD, with a significant increase in the time limit tolerated in a CWRET, and a decrease in dyspnea, ventilation and dynamic hyperinsuflation9, 10.
Oxygen delivery systems frequently used in PR are nasal cannulas, Venturi mask (VM) and mask with Reservoir bag. Recently, the market has incorporated a new system of oxygen delivery, the high-flow nasal cannula (HFNC), which is different from previous systems in that it provides heated and humidified gas, with higher flow (20 to 60 L/m) and a fraction of inspired oxygen (FiO2) of up to 100%. The HFNC allows for a decrease in oxygen dilution and the dead space of the airway; it generates positive pressure, improves oxygenation, reduces the respiratory rate and contributes to greater comfort11-14. The study of Cirio et al15 was the only published study that had evaluated the efficacy of the HFNC in exercise capacity tests and training. It was a crossover, randomized pilot study that compared the results of CWRETs using HFNC vs. VM in 12 patients with severe COPD and ventilatory limitation, with and without supplemental oxygen requirement during exercise. The limit time (Tlim) tolerated during the CWRETs was longer when using the HFNC (46% ± 36%). The patients showed less dyspnea and fatigue in the lower limbs; and greater oxygen saturation was observed at isotime15. The decrease in oxygen dilution, the reduction in the dead space and the generation of positive pressure produced by the HFNC could reduce dynamic hyperinflation and work of breathing in patients with COPD and could positively impact on the results of exercise capacity evaluations. This could allow us to provide training with higher intensity and longer duration, producing better results.
Although this research group commonly uses the HFNC for inpatients with acute respiratory failure and patients on the emergency list for lung transplantation during exercise, we have not yet analyzed if this oxygen delivery system is better than those frequently used in PR of patients with chronic respiratory diseases.
Our hypothesis was that the use of the HFNC would improve the exercise capacity during the IETs and CWRETs when compared to the use of VMs in patients with COPD requiring supplemental oxygen.
The main objective was to compare the maximum speed (MS) reached in an IET and the Tlim tolerated in a CWRET using two different systems for supplemental oxygen delivery: HFNC vs. VM. The secondary objectives were to compare the dyspnea and fatigue of the lower limbs, oxygen saturation (SpO2), heart rate (HR), respiratory rate (RR), and blood pressure (BP) during the different tests, and also to determine the comfort related to the use of the HFNC during exercise.
Materials and methods
Population: We carried out a crossover, randomized clinical trial including COPD patients admitted to the PR Program of the Hospital Italiano de Buenos Aires (Argentina), between April, 2016 and January, 2017. Individuals were invited to participate in the study if they satisfied the following inclusion criteria: diagnosis of COPD confirmed by spirometry in accordance with the GOLD16 criteria, indication of long term oxygen therapy (LTOT)17 or decrease in SpO2 below 88% during the Six Minute Walk Test (6MWT), and clinical stability of the disease defined as four weeks previous to starting the study not requiring antibiotics or oral corticosteroids. We excluded patients with neuromuscular, osteoarticular or cardiologic alterations that prevented or contraindicated the performance of the tests and those with respiratory
diseases other than COPD (for example, pulmonary fibrosis). We excluded patients showing any adverse event during the performance of the tests that would have forced us to stop the evaluation18 or any acute exacerbation of COPD leading to hospital admission or drug modification during the period of the study.
The project followed the World Medical Association Declaration of Helsinki, and all the patients signed the informed consent. The protocol was approved by the Ethics Committee on Research Protocols (known by its Spanish acronym, CEPI) No. 2719, together with the Board of Directors of the Hospital Italiano de Buenos Aires, and was registered at ClinicalTrials.gov, registration number NCT02858960.
In order to calculate the sample, we chose the Tlim of the CWRET as the main variable of the study, for being sensitive to consecutive changes made to interventions. Assuming a standard deviation of 184 and a minimal detectable difference of 100 seconds, according to previous literature19, 20, with an alpha error probability of 0.05 and a beta error of 0.20 (two-tailed), using paired measurements (repeated in each group), and assuming 20% loss to follow-up, we calculated a sample size of 28 participants in order to detect a minimum change of 100 seconds in the Tlim tolerated in the CWRET with the two oxygen delivery systems. The sample size was calculated using the GRANMO21 program.
Each participant included in the study attended five visits, separated by a period of 48 to 72 hours, to allow time for physical recovery. Figure 1 shows the tests that were conducted and the order in which they were put into practice.
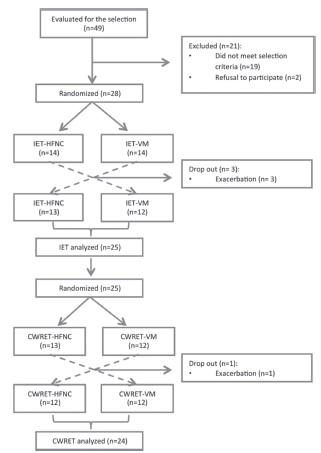
Analyzed parameters: In the first visit we recorded the demographic and clinical characteristics of the patient: age, sex, height, weight and use of Long Term Oxygen Therapy (LTOT). We calculated the Body Mass Index (BMI); carried out a spirometry using the Spirodoc® (MIR) spirometer, according to the standardization of the American Thoracic Society/European Respiratory Society (ATS/ERS)22; we administered the St George Respiratory Questionnaire (SGRQ)23 and conducted a 6MWT in accordance with the ATS criteria24. During the following visits, we conducted the IETs and, one member of the research team who subsequently, the CWRETs, according to the results of the randomization. One member of the research did not intervene in the participant selection process or the tests carried out the randomization of the order in which the tests were to be conducted with each oxygen delivery system (HFNC and VM), taking into account randomized numbers generated with the Stata program in blocks of 6, 8 and 10, then keeping them in closed envelopes numbered consecutively. The other team members were in charge of selecting the participants and the tests. Due to the type of intervention, neither the evaluators nor the patients could be blinded.
IET and CWRET evaluation technique: For the IETs and CWRETs with HFNC we used the AIRVOTM 2 system and OptiflowTM cannula (Fisher&Paykel Healht Care) with 50 L/m flow and 40% FiO2. For the IETs and CWRETs with VM (Hsinner), we also used 40% FiO2. Before starting with each test, the patients remained at rest for ten minutes, while the following parameters were recorded: SpO2, HR, RR, BP, dyspnea and fatigue using the modified Borg scale from 0 to 10 (Borg-Dyspnea and Borg-Fatigue) and the global comfort sensation using a Visual Analog Scale (VAS) from 0 to 10 (VAS - Comfort).
Incremental load tests: During the IETs, the patients walked on a treadmills, starting at 3 km/h with a 0.5 km/h increment every minute. Before each speed modification, we recorded SpO2, HR, dyspnea and fatigue. The reasons for ending the test were the patient’s desire to stop due to dyspnea or fatigue symptoms and the reasons to interrupt exercise tests for patients with pulmonary diseases18. After the test was finished, we registered the MS achieved by the patient, in km/h. This parameter corresponds to the speed of the last complete minute, and constitutes the main result of the test and one of the main variables of the study. We also recorded the SpO2, HR, RR, and BP, dyspnea, fatigue and global comfort. The same variables were recorded after three minutes of recovery.
Constant load tests: During the CWRETs, the patients walked at a constant speed, corresponding to 90% of the MS achieved during the IET with VM (used in this protocol as reference). The same variables as in the IETs were recorded every minute. The reasons for interrupting the test were the same as those previously mentioned plus the fact of reaching a 30-minute walk. After the test was finished, we recorded the Tlim in seconds, corresponding to the maximum time during which the patient was able to tolerate the test, considered together with the test main result as another main variable of the study. We also recorded the variables of physiological response to exercise mentioned in the IET.
Statistical analysis: We compared the physiological response variables according to the different devices of oxygen delivery (HFNC or VM) and for each of the exercise protocols (IET and CWRET) using a two-tailed, paired t test or a Wilcoxon test, according to data distribution. A value of p<0.05 was considered as statistically significant. The statistical analysis was conducted using STATA 12.1 (StataCorp, College Station, TX, USA). Sample characteristics and results variables are expressed as mean and standard deviation (SD), median and percentile 25-75 (P25-P75) or frequencies and percentages, according to variable distribution.
Results
During the study period, 49 patients with COPD were admitted to the PR Program. 14 patients did not meet the inclusion criteria, for they did not require supplemental oxygen during exercise. 5 patients were excluded due to osteoarticular problems, and 2 individuals refused to participate in the study. So, 28 patients were included in the study. Figure 1 details the flow of participants (see above).
56% of the participants were men, with a mean age of 69 years (SD 10) and a mean FEV1 theoretical percentage of 44% (SD 19). The demographic and clinical characteristics of the sample are described in Table 1. As observed in Table 2, there were no clinically relevant differences in the physiological variables of any of the groups at basal IET.
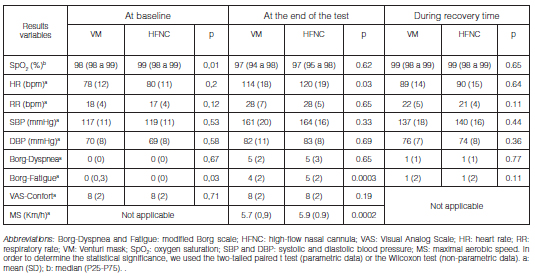
The MS obtained was significantly greater when the participants used HFNC, rather than VM (5.9 vs. 5.7 km/h; p = 0.0002); which implies a difference of 0.26 km/h (95% CI: 0.14 to 0.38) favorable for the HFNC.
When comparing at isoworkload (last complete speed level in which both incremental tests coincide), we observed less dyspnea (2.8 vs. 4.4; p = 0.0011) when using the HFNC, representing a difference of -1.6 (95% CI: 2.4 to -0.8) (Table 3).
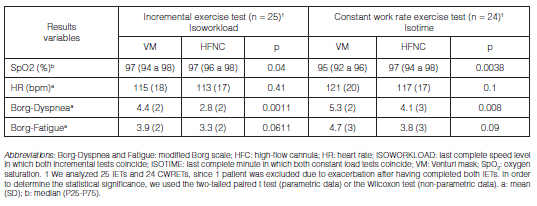
Figure 2 represents the mean differences (95% CI) in SpO2 (diff SpO2 = SpO2 HFNC - SpO2 VM) in favor of the HFNC, in comparison to VM in the IETs. In the same way, we observe the mean differences between dyspnea scores according to the modified Borg scale. As can be observed, the use of the HFNC resulted in an increase in SpO2 and a decrease in dyspnea at each instant measured during the tests, with predominance at isoworkload.
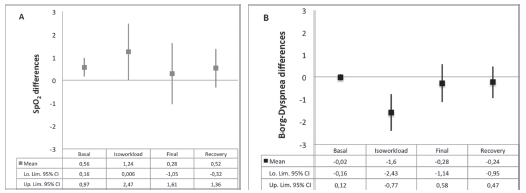
The CWRETs were conducted with a mean intensity of 5 km/h (SD 0.8), that corresponded to 90% of the MS achieved during the IETs with VMs. As can be observed in Table 4, there were not clinically relevant differences, either, in the physiological variables of any of the groups at basal CWRETs.
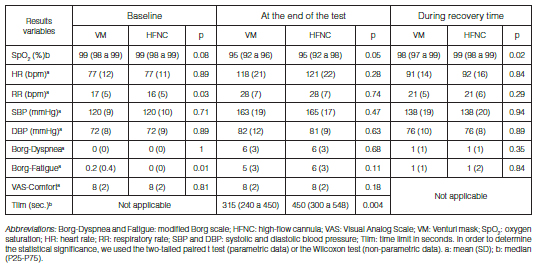
The Tlim during the CWRETs increased significantly with the use of the HFNC, in comparison with the VM (450 vs. 315 seconds; p = 0.004), representing a difference of 101 seconds in favor of the HFNC (95% CI: 40 to 161 seconds), surpassing the minimum clinically significant difference (MCSD) described for this test8.
Just like the IETs, at the isotime comparison (last complete minute in which both CWRETs coincide), patients claimed to have less dyspnea with the use of the HFNC (4.1 vs. 5.3; p = 0.008), representing a difference of -1.2 (95% CI: -2.1 to -0.3) with the VM, which is a statistically significant difference. We also confirmed a statistically significant difference of 2.3% (95% CI: 0.5 to 4.1) in the SpO2 (97 vs. 95%; p = 0.0038), without changes in the rest of the variables that were analyzed (Table 3).
No clinically relevant differences were observed in the physiological variables after the tests were finished or during recovery (Table 4).
Figure 3 represents the mean differences in SpO2 and dyspnea scores wich favor HFNC when comparing the VM to HFNC in the CWRET. Just like the IETs, the use of the HFNC resulted in an improved of the SpO2 and a decrease in dyspnea in each measurement, with the greatest difference at isotime.

Finally, when assessing the comfort according to the VAS, no statistically significant differences were observed in any of the tests or with any of the oxygen delivery device (see Tables 2 and 4 above). There was a mean VAS-comfort of eight points for both devices, where 10 is the maximum comfort score, with mean differences between the use of the HFNC and the VM of -0.2 (95% CI: -0.3 to 1.5) during the IETs and of 0.7 (95% CI: -0.3 to 1.7) during the CWRETs.
The patients did not present any adverse event associated with the tests or the use of either oxygen delivery devices. Also, furthermore none of the patients achieved the maximum time (30 minutes) in the CWRETs.
Discussion
To the best of our knowledge, this study is the first to analyze the influence of HFNC on exercise capacity during IETs and CWRETs through a randomized study. In our study we documented the fact that, in patients with COPD requiring supplemental oxygen, important benefits were attained with the use of HFNC, such as an increase in aerobic capacity and an increase in intensity during the IETs and the Tlim tolerated in the CWRETs, as well as the decrease in dyspnea and improvement of SpO2. Not surprisingly, there weren’t any adverse events during the tests. No complications were observed, either, associated with the use of oxygen delivery devices, and both systems were equally comfortable. This study is the first one to document that the use of the HFNC during exercise is safe and comfortable.
Probably one weakness of our study could be the inability to blind both patients and evaluators. The volunteers were aware at all times of the device with which they were being evaluated; consequently, the subjectivity of each patient could have influenced the aerobic exercise, the symptoms, or the feeling of comfort. It was technically impossible also to prevent the evaluators from knowing which device was being used in every test, and this might have also influenced the tests. But, when analyzing the results of the physiological variables measured through objective methods such as HR, SpO2, BP and RR, it could be clearly observed that these variables had a similar behavior after finishing the tests, showing that the effort made by the patients was similar regardless of the oxygen delivery method that was used.
In our study we selected a relative intensity of 90% of the MS obtained in the IET, as recommended that the CWRETs are conducted at high intensities to detect post-intervention changes8. Although the ERS (European Respiratory Society) recommends intensities between 75 and 80% when the tests are conducted in cycle ergometers, we chose a higher intensity because, according to our experience in CWRETs performed in treadmills, a great percentage of the patients complete 30 minutes when exercising at a intensity of 90% and this would not allow us to detect changes according to the oxygen delivery device that is being used. The intensity selected for the CWRETs caused 73% of the tests to last from 180 to 480 seconds (Tlim), an optimum time recommended by the ERS8.
In order to allow for the physical recovery of the patients, recovery times between one test and another in this study ranged from 48-72 hours. Such recovery times were longer than those in other studies9, 25 where the time between tests ranged from 30 to 90 minutes. Although the total duration of the study opened a longer time window for potential disease exacerbations, we believe it is necessary to have longer recovery times in order to optimize performance during the tests. Despite what has been described, the percentage of patients excluded from the study due to worsening of their condition was below the expected 20%.
With reference to the results of previous studies9, 25 which documented that the use of supplemental oxygen increases the capacity to exercise and that there is a greater benefit when a higher FiO2 is administered, we decided to use the same FiO2 in both devices in order to neutralize the effect of the FiO2 and discriminate the effects of the high-flow, which is present only in one of the devices.
When we compared our results with the only work within this line of research that was published by Cirio et al15, were we could observe similarities both in the FiO2 and flow used with the HFNC (40% FiO2 and 50 L/m flow in our study vs. mean of 44% ± 0.11 and 58.7 L/m flow in Cirio’ s study). Although the FiO2 in our study was equal for all the patients, instead of being titrated particularly for each one of them, as had been done by Cirio et al, we clarify there is no consensus about oxygen titration during the tests performed; and that the selected FiO2 (40%) was enough to avoid the interruption of the tests caused by a drop in SpO2 below 80%.
In our study, the Tlim of the CWRET increased from 315 seconds with the use of VM to 450 seconds with the use of the HFNC, a statistically significant difference exceeding the MCSD of 100 seconds8. Our results are consistent with those observed by Cirio et al, who documented a mean increase of 109 seconds.
Another comparable result as seen in Ciro et al15 was the higher SpO2 observed during the CWRETs when using the HFNC (97% with HFNC vs. 95% with VM; and 92% with HFNC vs. 89% with VM). The increase in SpO2 could be explained by the use of high flow to administer the oxygen (50 L/m). This flow is higher than the maximum possible inspiratory flow of the patient (MVV) calculated for each patient according to their FEV1, with a median of 30 L/m, making it possible to maintain a constant FiO2, wich differs from the VM.
Also, a significant decrease in dyspnea was observed at isotime (mean of 4.1 with HFNC vs. 5.3 with VM). Although there is not a MCSD for this variable at isotime during the CWRET, the improvement of one or more points in the Borg scale is considered relevant8. The difference in dyspnea observed in our study was clearly lower than the one documented by Cirio et al15 (5.5 with HFNC vs. 10 with VM). This difference might have been caused by the fact that the symptoms claimed by our patients during and after the tests were much less than those reported by Cirio et al. These discrepancies could be due to cultural issues related to the assessment of the symptoms, since it is not common in our practice to have patients claiming the maximum values of the modified Borg scale during exercise or training tests, and also to the fact that the condition of the patients in the Cirio et al study was slightly more severe than the cases included in our study.
It wasn’t possible to compare the comfort of the HFNC with the Cirio study, given that this device was not assessed in such study. The basal and intratest comfort of the present study was 8/10, according to the VAS.
As previously mentioned, our study was the first to assess the effects of HFNC during IET. The results were similar to those observed during the CWRETs, showing an increase in the exercise capacity and a decrease in dyspnea when using the HFNC, in comparison to the VM. Although no MCSDs have been documented in the MS, it is common to find mild changes after a training program. The magnitude of the change in the MS documented in this study was lower than the change observed in the Tlim of the CWRET. These differences were as expected, since the CWRETs are more sensitive than the IETs in detecting changes in the post-intervention exercise capacity. We documented a 33% increase in the Tlim of the CWRETs in most non-pharmacologic interventions, and a mean of 11% in the peak oxygen consumption in the post-rehabilitation IETs8.
The results of the present study show that the HFNC allows COPD patients who require supplemental oxygen to tolerated higher load intensity and longer duration during exercise tests, with better SpO2 and less dyspnea. These results could be due to the effect the HFNC has in decreasing oxygen dilution, the reduction of dead space and generation of positive pressure in the airway, causing a decrease in ventilatory requirement, dynamic hyperinflation and, consequently, work of breathing. A previous study9 assessing the effects of oxygen administration showed a correlation between the decrease in dyspnea at isotime during the CWRETs and the increase in the inspiratory capacity (indirect measurement of dynamic hyperinflation); so, we can assume that the use of the HFNC caused a decrease in hyperinsuflation, since less dyspnea was observed.
The results of the exercise tests of the PR program are used to plan the individual training sessions of each patient. According to the information of the present study, the use of the HFNC in this population could allow patients to train at a higher intensity and for a longer time with less dyspnea, thus obtaining better training results, though additional studies are needed in order to prove this hypothesis.
The fact that the sample of the study is the typical population of COPD patients who partake in a pulmonary rehabilitation program and that the devices are widely known and becoming increasingly available gives our study great external validity and enforceability.
Conclusion
We conclude that the delivery of supplemental oxygen using HFNC, in comparison with VM, improved the exercise capacity in COPD patients, this is shown by the increase in the MS achieved in the IETs and the Tlim tolerated in the CWRETs. We also observed an increase in the SpO2 and a lower intensity of dyspnea symptoms during the tests with HFNC lastly, the VM and HFNC show to be equally comfortable.
Conflicts of Interest and Economic Considerations
The authors declare there is no any conflict of interest, commercial or work relationship with the brands mentioned in the protocol.
This study was co-financed by the Kinesiology Department Service of the Hospital Italiano de Buenos Aires and the 2016 Research Scholarship granted by the Argentinian Association of Respiratory Medicine (AAMR, for its acronym in Spanish).
Acknowledgement
To the Kinesiology Department Service of the Hospital Italiano de Buenos Aires, Argentina.
To the Board of Directors of the Hospital Italiano de Buenos Aires, Argentina.
To the Argentinian Association of Respiratory Medicine (AAMR) for granting the 2016 Research Scholarship.
1. Maltais F, Decramer M, Casaburi R, et al. ATS/ERS Ad Hoc Committee on Limb Muscle Dysfunction in COPD. An official American Thoracic Society/European Respiratory Society statement: update on limb muscle dysfunction in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2014; 189(9): e15-62.
2. Rabinovich RA, Vilaró J. Structural and functional changes of peripheral muscles in chronic obstructive pulmonary disease patients. Curr Opin Pulm Med. 2010; 16(2): 123-33.
3. Spruit MA, Singh SJ, Garvey C, et al. ATS/ERS Task Force on Pulmonary Rehabilitation. An official American Thoracic Society/European Respiratory Society statement: key concepts and advances in pulmonary rehabilitation. Am J Respir Crit Care Med. 2013; 188(8): e13-64.
4. Güell Rous MR, Díaz Lobato S, Rodríguez Trigo G, et al. Pulmonary rehabilitation. Sociedad Española de Neumología y Cirugía Torácica (SEPAR). Arch Bronconeumol. 2014; 50(8): 332-44.
5. Bolton CE, Blakey JD, Morgan MD; BTS Pulmonary Rehabilitation Guideline Development Group for the Standards of Care Committee for the BTS. The British Thoracic Society guideline on pulmonary rehabilitation in adults: your opinion is noted. Thorax. 2014; 69(4): 388-9.
6. Sívori M, Almeida M, Benzo R, et al. New argentine consensus of respiratory rehabilitation 2008. Medicina (B Aires). 2008; 68(4): 325-44.
7. Patessio A, Carone M, Ioli F, Donner CF. Ventilatory and metabolic changes as a result of exercise training in COPD patients. Chest. 1992;101 (5 Suppl): 274S-278S.
8. Puente-Maestu L, Palange P, Casaburi R, et al. Use of exercise testing in the evaluation of interventional efficacy: an official ERS statement. Eur Respir J. 2016; 47(2): 429-60.
9. O’Donnell DE, D’Arsigny C, Webb KA. Effects of hyperoxia on ventilatory limitation during exercise in advanced chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2001; 163(4): 892-8.
10. Nonoyama ML, Brooks D, Lacasse Y, Guyatt GH, Goldstein RS. Oxygen therapy during exercise training in chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2007; (2): CD005372.
11. Frat JP, Thille AW, Mercat A, et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015; 372(23): 2185-96.
12. Dysart K, Miller TL, Wolfson MR, Shaffer TH. Research in high flow therapy: mechanisms of action. Respir Med. 2009; 103(10): 1400-5.
13. Spoletini G, Alotaibi M, Blasi F, Hill NS. Heated Humidified High-Flow Nasal Oxygen in Adults: Mechanisms of Action and Clinical Implications. Chest. 2015; 148(1): 253-61.
14. Nishimura M. High-flow nasal cannula oxygen therapy in adults. J Intensive Care. 2015; 3(1): 15.
15. Cirio S, Piran M, Vitacca M, et al. Effects of heated and humidified high flow gases during high-intensity constant-load exercise on severe COPD patients with ventilatory limitation. Respir. Med. 2016; 118: 128-132.
16. Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global Strategy for the Diagnosis, Management and Prevention of Chronic Obstructive Lung Disease 2017 Report: GOLD Executive Summary. Respirology. 2017; 22(3): 575-601.
17. Hardinge M, Suntharalingam J, Wilkinson T; British Thoracic Society Guideline update: The British Thoracic Society Guidelines on home oxygen use in adults. Thorax. 2015; 70(6): 589-91.
18. ERS Task Force, Palange P, Ward SA, Carlsen KH, et al. Recommendations on the use of exercise testing in clinical practice. Eur Respir J. 2007; 29(1): 185-209.
19. Cooper CB, Abrazado M, Legg D, Kesten S. Development and implementation of treadmill exercise testing protocols in COPD. Int J Chron Obstruct Pulmon Dis. 2010; 5: 375-85.
20. Casaburi R, Kukafka D, Cooper CB, Witek TJ Jr, Kesten S. Improvement in exercise tolerance with the combination of tiotropium and pulmonary rehabilitation in patients with COPD. Chest. 2005; 127(3): 809-17.
21. Marrugat J, Vila J, Pavesi M, Sanz F. Estimation of the sample size in clinical and epidemiological investigations. Med Clin (Barc). 1998; 111(7): 267-76.
22. Laszlo G. Standardisation of lung function testing: helpful guidance from the ATS/ERS Task Force. Thorax. 2006; 61(9): 744-6.
23. Jones PW, Quirk FH, Baveystock CM. The St George’s Respiratory Questionnaire. Respir Med. 1991;85 Suppl B: 25-31; discussion 33-7.
24. Holland AE, Spruit MA, Troosters T, et al. An official European Respiratory Society/American Thoracic Society technical standard: field walking tests in chronic respiratory disease. Eur Respir J. 2014; 44(6): 1428-46.
25. Dewan NA, Bell CW. Effect of low flow and high flow oxygen delivery on exercise tolerance and sensation of dyspnea. A study comparing the transtracheal catheter and nasal prongs. Chest. 1994; 105(4): 1061-5.