Autor : Bertolotti Alejandro Mario1,2, Gilbert Mónica2, Gómez Carmen Beatriz1, Absi Daniel Oscar2, Ossés Juan Manuel1, Favaloro Roberto René1,2
1Lung Transplantation Service of the Hospital Universitario FundaciĂłn Favaloro, Argentine 2 Cardiovascular and ECLS Service, HUFF, Argentine
Correspondencia : abertolotti@ffavaloro.org
Abstract
Introduction: Several studies have demonstrated the benefits of extracorporeal life support in critically ill patients with cardiorespiratory diseases. The purpose of this study was to evaluate the preliminary experience of the use of extracorporeal life support in patients with advanced pulmonary disease on the waiting list for lung transplantation.
Materials and Methods: We conducted a retrospective and descriptive analysis of patients who received extracorporeal life support as a bridge to lung transplantation or to lung transplantation candidacy between August 2010 and July 2015. Results were analyzed according to: exclusion from the waiting list, transplant candidates and number of transplants performed. We described complications and causes of mortality, and duration of extracorporeal life support, mechanical respiratory assistance and post-transplant hospitalization.
Twenty-three patients were included, with a mean age of 36 ± 17 years; 61% were female. The most common diagnoses were cystic fibrosis (34.8%), idiopathic pulmonary fibrosis (30.4%) and secondary pulmonary fibrosis (13.0%).
Results: Mean time on extracorporeal life support was 14.4 ± 11.7 days; veno-venous support was implemented in 14 patients, veno-arterial support in 4 cases, arterio-venous in 3, and veno-arterio-venous in 2. Eight patients did not qualify as candidates for transplantation and were excluded from the waiting list. 15 patients were accepted as a bridge for lung transplant; 8 of them died while receiving extracorporeal life support, and 7 were transplanted. After the transplantation, one patient died and 6 were discharged from the hospital with more than one year survival.
Conclusion: In our series, 46.7% of candidates had access to lung transplantation under extracorporeal life support. The implementation of an effective extracorporeal life support program as a bridge to lung transplantation requires the access to new technologies and the development of the Center’s team experience.
Key words: Extracorporeal life support; Extracorporeal membrane oxygenation; Bridge to transplantation; Lung transplantation.
Introduction
The extracorporeal life support (ECLS) has been used for treating severe respiratory failure and serious cardiogenic shock for more than four decades1. The extracorporeal membrane oxygenation (ECMO) is an advanced invasive technique of partial or total support of the heart-lung function, but due to mechanical, infectious and bleeding complications, its use in the clinical practice is limited. However, in 2009, after the H1N1 influenza A epidemic associated with adult respiratory distress syndrome (ARDS), the success of the support provided by the ECMO in comparison with the conventional mechanical ventilatory assistance2 became evident. The CESAR study (Conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure) for the comparison of the use of conventional mechanical ventilation with ECMO in adults with severe respiratory failure showed a significant benefit in the survival of patients on ECMO, in comparison with the control group. These results lead to the daily implementation of the ECMO in more than 200 centers at international level3, 4.
In accordance with the ELSO (Extracorporeal Life Support Organization), these devices were designed as a tool to temporarily provide support to patients with respiratory failure and/or hemodynamic instability, allowing for suitable oxygenation and homeostasis during a period of time necessary for the recovery or the definitive treatment for the underlying disease5. For the last two decades, the use of ECLS in patients with advanced end-stage pulmonary disease as a bridge to definitive treatments has been occasional. At the present time, almost 1.8% of patients on the waiting list who have access to lung transplantation are under some modality of ECLS6.
The purpose of this study was to evaluate the preliminary experience of the use of ECLS as a bridge to lung transplantation in patients who are refractory to the conventional medical treatment in a single center.
Materials and Methods
Population: We conducted a retrospective and descriptive analysis of the clinical records of all the patients who received ECLS as a bridge to lung transplantation or to transplant candidacy between August 2010 and July 2015, in a single center. All of the patients were active on the waiting list for lung transplantation in accordance with the regulations of the Unique Central National Institute Coordinator of Ablation and Transplant (INCUCAI, according to its acronym in Spanish), and were registered in the National System of Procurement and Transplantation Information of the Argentine Republic (SINTRA, according to its acronym in Spanish). (https://sintra.incucai.gov.ar/)
Definitions: We considered as “bridge to lung transplantation” the cases in which the patient’s initial condition did not represent an absolute contraindication to transplantation; and the cases considered as “bridge to lung transplantation candidacy” were those in which the patient’s initial condition when the support was being implanted was an absolute contraindication to receive a transplant and was used as a salvage treatment. We considered as “treatment futility” the situations where, despite the use of the proper ECLS, the associated comorbidities represented an absolute contraindication to transplantation, in which case the INCUCAI was informed about the patient’s definitive removal from the waiting list. In those cases we decided, together with the patients’ direct relatives, to remove their ECLS and establish end-of-life palliative support measures.
Indications: We indicated ECLS in patients with advanced respiratory failure (pulmonary fibrosis, cystic fibrosis, bronchiectases) previously evaluated for lung transplantation and registered on the waiting list, who developed severe hypoxemia and/or hypercapnia refractory to step-care conventional treatments: High flow oxygen therapy with reservoir mask, noninvasive ventilation (NIV) with different interfaces, NIV with high oxygen flow and mechanical respiratory assistance (MRA) with orotracheal intubation. The criterion for indicating ECLS in patients on the waiting list for lung transplantation is the one recommended by the ELSO Guidelines, versions 1.1 and 1.3 (1:1 April 2009 and Version 1:3 December 2013, respectively; Ann Arbor, MI. https://www.elso.org/resources/guidelines.aspx), which establish that the requirement for invasive ventilation in patients awaiting lung transplantation is an indication to begin with ECLS treatment.
In these patients, we used the arterio-venous (AV) ECLS modality for ventilatory support (hypercapnia) or the veno-venous modality (VV) for respiratory support (hypoxemia with or without hypercapnia).
ECLS was indicated in patients who developed cardiogenic shock (pulmonary hypertension and right ventricular failure) whenever it was impossible to compensate the hemodynamic condition with endovenous inotropic support (milrinone at maximum dose), pulmonary vasodilators and/or endovenous diuretics to obtain a negative hydric balance. For the use of ECLS in these cases, the venous-arterial (VA) or venous-arterio-venous (VAV) modalities were chosen.
Management of patients on ECLS: clinical, circuit and membrane, and laboratory parameters were monitored periodically. All of the patients received anticoagulant treatment with unfractionated heparin intravenously administered at an initial dose of 50 UI/kg, and follow-up with initial infusion of 5-10 UI/kg/h, titrating the dose until an activated partial thromboplastin time (APTT) of 60 to 80 seconds was obtained. The circuit was systematically monitored for detecting thrombi or fissures. The functionality of the ECMO was assessed through changes in the transmembrane gradient and the values of postmembrane arterial gases.
No protocol prophylaxis with antimicrobials or antifungals was carried out; only germ-specific treatments were indicated in patients who had developed infectious processes identified by cultures during the support, or against germs previously identified by microbiological isolators such as colonizers (for example, septic pulmonary diseases such as cystic fibrosis and non-cystic fibrosis bronchiectasis) if no microorganism growth was obtained in the cultures.
Once the ECLS had been installed, if the clinical condition was adequate (patient hemodynamically and gasometrically stable, with no organic failure), progression to spontaneous ventilation was prioritized in patients under MRA, using minimum levels of sedation and analgesia and avoiding the use of neuromuscular blockade. In these patients, we used a protective ventilation strategy every time it was possible (FiO2 at 0.4 and maintenance of the plateau pressure < 25 cmH2O). Early tracheotomy was implemented. In selected cases, we considered extubation (vigil and/or ambulatory ECMO) with physical rehabilitation and ambulation measures.
Statistical Analysis: the demographic characteristics, the number of transplants performed and their survival, mortality of patients under ECLS and post-transplant mortality were analyzed. We compared the gasometry before and after the implementation of the support. Continuous variables with normal distribution were expressed as mean and standard deviation, and variables with non-Gaussian distribution, as median and confidence intervals. Categorical variables were detailed as proportions and frequencies. Quantitative variables were compared with the Wilcoxon-Mann Whitney method. A value of p < 0.05 was considered a significant difference. The SPSS statistical program, version 17 was used.
Results
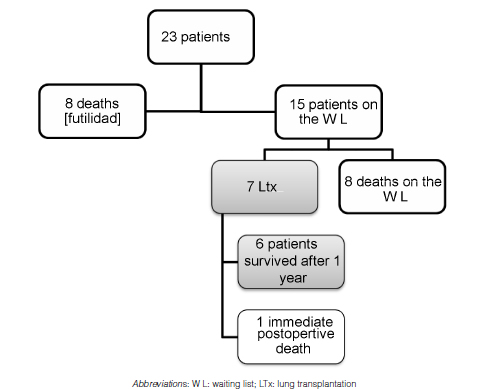
23 patients were included. They received an ECLS strategy as a bridge to lung transplantation or to lung transplantation candidacy. Figure 1 shows the final destination of the different subgroups analyzed in this study. Table 1 summarizes the demographic characteristics, type of support and evolution of the 23 cases included in the study. The mean age was 36 ± 17 years; 61% were female. The diagnoses were: cystic fibrosis (n=8; 34.8%), idiopathic pulmonary fibrosis (n=7, 30.4%), secondary pulmonary fibrosis (n=3; 13.0%), post-lung transplantation bronchiolitis obliterans (n=2; 8.7%), group 1 pulmonary hypertension (n=2; 8.7%) and bronchiectases (n=1; 4.3%).
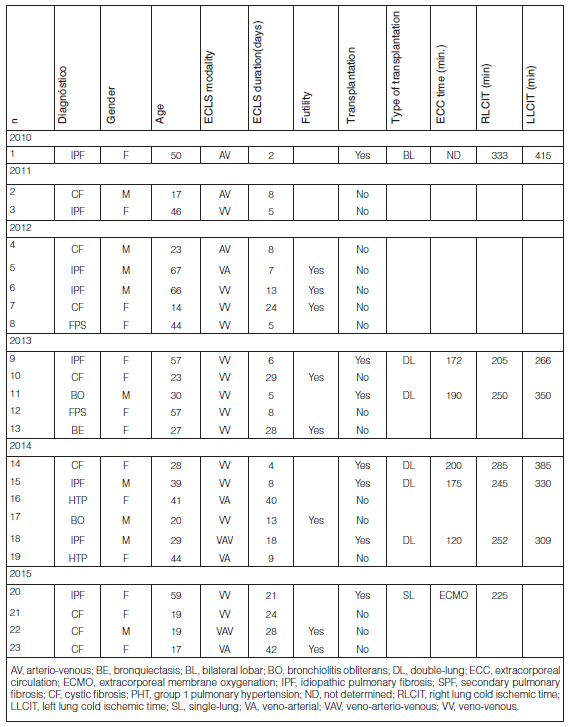
The ECLS was installed with the VV modality in 14 patients (60.8%), VA in 4 (17.4%), AV in 3 (13.0%) and VAV in 2 (8.7%). Cannulation sites were: for the VV modality, femoral vein-jugular vein (n= 12) and jugular vein with dual lumen cannula (Avalon®) (n= 2); for the VAV modality, femoral vein and artery plus jugular vein (n= 2); femoral vein and artery (n=3) for the AV modality and; at last, for the VA modality, we used femoral vein and artery (n= 2), jugular vein and femoral artery (n= 1) and veno-arterial central cannulation (n= 1). Access sites were obtained by percutaneous puncture with Seldinger technique in 20 patients and by surgical dissection in the 3 remaining patients. The correct placement of the dual lumen cannula at the superior vena cava was confirmed by radioscopic control during the cannulation procedure.
The centrifugal pumps used in the ECLS circuits were: 18 Rotaflow® (Maquet Getinge Group), 2 Bio- Medicus® (Medtronic). The membranes that were used were mostly polymehtylpentene (18 Quadrox®, Maquet Getinge Group and 3 iLA Novalung®) and 2 Affinity® (Medtronic) membranes.
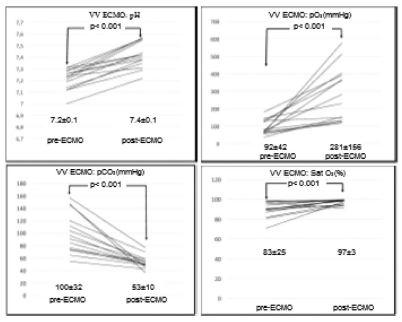
Figure 2 shows changes in gasometry values before and after the implantation of the ECLS with the veno-venous modality.
Total mean time of ECLS was 14.4 ± 11.7 days in all the patients.
Futility under ECLS: 8 of the 23 patients (34.7%) showed absolute contraindications for the transplantation, despite the use of the ECLS, so we considered those cases as treatment futility. All of these patients were disconnected from the support and died under end-of-life palliative care. The contraindications that motivated futility were: stroke in two cases and sepsis with multiorgan failure in 6 patients (septic pulmonary disease: 5 cystic fibrosis; 1 non-cystic fibrosis bronchiectasia); bronchiolitis obliterans post-lung transplantation in 1 case and idiopathic pulmonary fibrosis in a 67-year-old patient.
Bridge to lung transplantation with ECLS: 46.7% (n= 7) of the remaining 15 patients had a successful bridge to transplantation within an ECLS mean time of 11 ± 7 days (min. 2 – max. 21). One of these patients had previously received a lung transplant for advanced cystic fibrosis and had developed chronic graft failure (bronchiolitis obliterans) at 4 year-survival, thus receiving a lung retransplantation under pre-transplant support with ECLS.
Six out of 7 transplanted patients (85.7%) survived after 30 days (Figure 1) and were discharged. One patient died in the operating room during the implantation procedure due to serious lung injury and cardiac arrest refractory to resuscitation measures. The lung transplantation was carried out with extracorporeal circulation (ECC) pump in 6 cases and with intraoperative ECMO in 1 patient. Six patients (85.7%) received a double-lung transplantation (1 of them was a retransplant) and one patient received a single-lung transplantation.
Post-implantation complications were: grade 3 serious primary graft failure (n= 3), nondialytic renal failure (n= 3), empyema (n= 3), pneumothorax and pneumomediastinum (n= 2), bacterial pneumonia (n= 2), re-operation due to bleeding (n= 2).
The mean hospitalization stay since admission before the transplantation until discharge was 60 ± 19 days. Mean MRA times, mean length of stay at the Intensive Care Unit (ICU) and total stay after the transplantation were 20 ± 12 days; 28 ± 15 days and 46 ± 27 days, respectively. The 6 patients who were discharged got through the first follow-up year with a 1 year survival of 86%.
Mortality with ECLS: eight patients (53.3%) died while they were on ECLS as a bridge to transplantation. The mortality causes were septic shock (n= 3), accidental baldness (n= 3) and bleeding complications (n= 2): retroperitoneal hemorrhage in one case and bleeding due to coagulopathy in the remaining case. The mean support time of these patients was 12 ± 11 days (min. 5 – max. 40).
Discussion
Lung transplantation is the effective treatment for advanced pulmonary disease. The worldwide shortage of donors and the increase in the number of waiting list patients make this population more vulnerable; also, once the patient is included in the waiting list, his/her mortality is higher in comparison with candidates for transplantation of other organs. On the other hand, the heterogeneity of the diseases eligible for lung transplantation in terms of prognosis, associated comorbidities and age groups (among other differences), poses a great dilemma regarding the fair and equal allocation of the lacking resource. In this context, regarding the lung transplantation waiting lists, changes have been globally introduced in the process of donor lung allocation. In the United States, for example, a standardized score system was implemented, the Lung Allocation Score (LAS), thus reducing the number, total time and mortality of patients on the waiting list7. With this interpretation of organ allocation, which began to spread throughout the world in 2005, patients with MRA received high scores, thus allowing them to move up on the list and have greater possibilities to receive the transplant. However, the dependency on the MRA, the severity of these critically ill patients, and the poor survival rates one year post-transplantation determined (though did not prohibit) their inclusion in transplantation programs in some centers8.
In our country, since 2010, a new cadaveric donor lung allocation system has been used. This system includes four categories (Emergency, Urgency A, Urgency B, Elective), based on clinical condition, underlying disease and the need for oxygen therapy, noninvasive ventilation, mechanical respiratory assistance and extracorporeal support, in the expectation of generating equity and opportunities for patients on the waiting list with high mortality rates and with better results shown after the transplantation9.
Despite the use of mechanical ventilation and conventional medical treatment, there is still a group of patients refractory to this type of treatment, and their progressive clinical impairment carries a very bad prognosis in the short term, with 100% mortality if they don´t have access to a transplant. The need of critically ill patients to maintain homeostasis during their time on the waiting list was the reason to implement another type of vital support as a “bridge to transplantation”10.
After being used for more than four decades, it was after the year 2000, particularly after 2006 with the appearance of new technologies (polymethylpentene membrane oxygenation, more effective centrifugal pumps with less hemolysis and heparin-coated circuits) that the ECMO as a bridge to lung transplantation became interesting again. There is also the experience acquired with the ECLS during the H1N1 influenza A epidemic in 2009, after which the use of ECLS as a bridge to lung transplantation became more popular11.
In our Center, the lung transplantation program began in 1994. Since then, more than 350 lung and heart-lung transplants have been performed. The increasing experience obtained by the transplantation team throughout more than 20 years allows us to analyze the results of critically ill patients refractory to conventional medical treatment awaiting lung transplantation on ECLS between August, 2010 and July, 2015, taking into account during this period not only the country’s availability of a new ECLS technology, but also the new national categorization system of patients on the waiting list and the greater experience of the medical and paramedical staff.
During the period under examination, 128 lung transplantations were performed. 7 patients (5.4%) of this study who had previously been assisted with ECLS as a bridge to transplantation could be transplanted. The work of Gulack et al states that the percentage of patients with ECLS as a bridge to transplantation is 1.8%, a figure below the one found in our series6. This difference arises as a need of our lung transplantation service to offer greater opportunities to critically ill patients, driven by the low lung procurement rate in Argentina, in comparison with other countries with higher procurement rates per million inhabitants.
The ECLS was used in our Center for patients on the waiting list for lung transplantation who presented rapidly progressive impairment of their clinical status, requiring MRA in all the cases. In these cases with very bad prognosis in the short term, the chosen strategy was a bridge to transplantation, when no absolute contraindications had been identified, or a bridge to candidacy, for patients who presented comorbidities that contraindicated the transplant but could be potentially reversible: hemodynamic instability with inotropic and/or vasopressor support, active uncontrolled infection, renal failure or liver failure. Although the initial strategy was to offer opportunities to all 23 patients, there were 8 cases (34.8%) in which it was not possible to correct the comorbidities, so treatment futility was declared with ex professo culmination of the support and end-of-life care. The decision to declare futility represented a great emotional challenge for the treating group, and the experience that was gained will allow for the adjustment of ECLS indication to minimize these instances and also the development of communication strategies towards the family and medical staff for them to take the risk of facing this situation.
Lafargue published that 16.6% (6/36) of patients on the waiting list died before they could receive a viable organ12. In our initial experience with the use of ECLS as a bridge to transplantation, the percentage of patients without contraindications who died and could not receive the donor lung was 53.3% (n= 8/15), and the causes of death were related to mechanical complications (accidental decannulation) associated with anticoagulation (coagulopathy, retroperitoneal bleeding) and uncontrolled infection (sepsis with multiorgan failure). The first two causes of failure are directly related to the development of a learning curve involving all the medical staff. In the Lafarge series, 83.3% (n= 30/36) of patients were able to receive a transplant: 75% achieved survival 30 days post-transplantation and 60% one year post- transplantation12.
Mason et al achieved a successful bridge to lung transplantation in 56.9% (n= 29/51) of patients, with a subsequent survival of 72% after one month and 53% after one year13. Hoopes et al reported 97% survival 30 days post-transplantation, and 93% survival one year post-transplantation in 31 patients who had been treated with ECMO before the transplantation14. In our series, 7/15 patients (46.7%) received the lung transplant, with 30 days and 1 year post-transplantation survival of 86% and 86%, respectively. These figures are similar to the experience published by other centers.
The use of ECLS as a bridge to lung transplantation is associated with high perioperative morbidity and mortality that can be acceptable in the medium term; and in the long term, there is no difference between patients who received and those who did not receive ECMO as support before lung transplantation15. As more experience is gained, as observed in the Fuehner et al series, patients who were kept vigil during ECMO, that is to say, with assistance but awake, without sedation or orotracheal intubation, showed better survival rates compared to those who were receiving MRA at the moment of the transplantation (80% versus 50%, respectively)16. In three of the patients of our series we were able to use the ECLS without mechanical ventilatory assistance (vigil ECMO). We couldn’t implement an ambulation plan, but the possibility to be awake without ventilatory support allowed for lower general
muscular deconditioning, and particularly lower respiratory deconditioning. In order to draw conclusions about these statements, we need to have more cases managed with this strategy.
The remarkable differences in the results published between early and most recent experiences probably show improvements in the management and selection of both patients and technology. In this series, of the 7 patients who had a successful bridge to the transplant, we can focus on two periods at our Center: the first one (from August 2010 to December 2012) where 1/5 patients with ECLS (20.0%) was able to undergo the lung transplantation; and a more recent period (from January 2013 to July 2015) where 6/10 assisted patients (60.0%) could be successfully transplanted. This difference is related to the acquisition of new technologies as well as better training and greater experience of the medical staff in charge of these patients.
In our group of patients with ECLS as a bridge to transplantation, the underlying etiology was mostly cystic fibrosis and pulmonary fibrosis, similar to the series reported by Lafarge12. In our series, 5 out of 7 patients who were able to receive a transplant had pulmonary fibrosis, 1 had cystic fibrosis and 1 had bronchiolitis obliterans post-lung transplantation. Lang et al published a series of 5 patients who received a lung retransplant after having been assisted with vigil ECMO, whose results could be comparable to elective retransplants17.
The type of surgical resolution in our patients was mostly double-lung transplantation (6/7; 85.7%) with post-transplant morbidity and prolonged hospital stay. These variables partly reflect the difficulty in the post-surgical recovery of seriously ill patients who were weakened during the transplant waiting time.
Additional mortality risk factors should be considered in the selection of patients for lung transplantation in order to maximize survival. Yusen et al published a multivariate analysis of the potential mortality risks 1 year post-lung transplantation, identifying the baseline diagnosis (greater risk of interstitial pulmonary disease, sarcoidosis, non-cystic fibrosis bronchiectasias), retransplantation, previous stay at the ICU, pre-transplantation mechanical ventilation and need for dialysis, among others18. The main risk factors identified in the multivariate analysis of continuous risk factors of mortality after one year were: the age of recipients of more than 55 years old, the volume of the transplantation center below 35 transplants per year and the increasing levels of total bilirubin and creatinine before the transplant. It is important to underline that this analysis does not identify patients who were transplanted under ECLS18.
We believe the use of ECLS as a bridge to transplantation in centers with low- volume of transplants per year should be carefully considered. It is essential for the transplantation team to know the country’s average waiting times related to the procurement of dead donor lungs, since low procurement plus prolonged times limit the options for these patients, extending the time they receive support and increasing the risk of suffering complications. The transplantation team must have experience in the use of lung donors with expanded criteria and different surgical implantation techniques (such as lobar transplants) in order to increase these patients’ options. Also, it is necessary to create a rehabilitation program and use the suitable means to sustain these patients with ECLS keeping them vigil and ambulating (ambulatory ECMO), thus favoring earlier recovery possibilities after the transplant19.
Our work shows the following limitations: 1) the retrospective nature of the analysis, 2) the reduced number of patients, and 3) the heterogeneity of the technology applied to the ECLS, which complicate the fact of drawing accurate conclusions. However, it offers information about the feasibility of the use of ECLS in patients with diseases of a worse prognosis, such as cystic fibrosis, pulmonary fibrosis and primary and secondary pulmonary hypertension with right ventricular compromise, where mechanical ventilatory assistance is insufficient and damaging, because it alters the hemodynamic stability. The use of ECLS in these patients allows for an effective support for patients awaiting transplant.
The use of ECLS in patients with advanced pulmonary disease on the waiting list for lung transplantation who evolve without an adequate response to conventional treatments satisfies the need to offer the methodological tools necessary to keep these patients alive until they receive the definitive treatment, as for example, lung transplantation. The continuous evaluation of the clinical condition of a patient on ECLS as a bridge to lung transplantation is mandatory to confirm or reject the patient’s permanence
on the waiting list for transplantation. In our series, 46.7% of patients on the waiting list who met the transplantation criteria and were being assisted with ECLS had access to lung transplantation. These patients wouldn’t have been able to access the transplant without the “bridge to surgery” strategy, and would have otherwise died as a consequence of their end-stage chronic disease.
The implementation of an effective ECLS program as a bridge to lung transplantation requires the combination of access to new technologies, an adequate annual volume of lung transplants and greater experience in the management of these technologies by the medical staff in charge of these patients.
1. Hill J, De Leval M, Fallat R, et al. Acute respiratory insufficiency. Treatment with prolonged extracorporeal oxygenation. J Thorac Cardiovasc Surg 1972; 64: 551-562.
2. Combes A, Pellegrino V. Extracorporeal membrane oxygenator for 2009 influenza A (H1N1)-associated acute respiratory distress syndrome. Semin Respir Crit Care Med 2011; 32: 188-194.
3. Peek G, Mugford M, Tiruvoipati R, et al; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 2009; 374: 1351-63.
4. Del Sorbo L, Boffini M, Rinaldi M, Rainieri V. Bridging to lung transplantation by extracorporeal support. Minerva Anestesiol 2012; 78: 243-50.
5. Conrad S, Rycus P, Dalton H. Extracorporeal Life Support Registry Report 2004. ASAIO J 2005; 51: 4-10.
6. Gulack B, Hirji S, Hartwig M. Bridge to lung transplantation and rescue post-transplant: the expanding role of extracorporeal membrane oxygenation. J Thorac Dis 2014; 6: 1070-1079.
7. Egan T, Kotloff R. PRO/condebate: Lung allocation should be based on medical urgency and transplant survival and not on waiting time. Chest 2005; 128: 407-15.
8. Crotti S, Iotti G, Lissoni A, et al. Organ allocation waiting time during extracorporeal bridge to lung transplant affects outcomes. Chest 2013; 144: 1018-1025.
9. Resolución INCUCAI 385/10: Normativa Intratorácica. En: http//:www.incucai.gob.ar
10. Schmidt M, Pellegrino V, Combes A, Scheinkestel C, Cooper D, Hodgson C. Mechanical ventilation during extracorporeal membrane oxygenation. Critical Care 2014; 18: 203.
11. Chiumello D, Coppola S, Froio S, Colombo A, Del Sorbo L. Extracorporeal life support as bridge to lung transplantation: a systematic review. Critical Care 2015; 19: 19.
12. Lafarge M, Mordant P, Thabut G, et al. Experience of extracorporeal membrane oxygenation as a bridge to lung transplantation in France. J Heart Lung Transplant 2013; 32: 905-913.
13. Mason D, Thuita L, Nowicki E, Murthy S, Pettersson G, Blackstone E. Should lung transplantation be performed for patients on mechanical respiratory support? The US experience. J Thorac Cardiovasc Surg 2010; 139: 765-73.
14. Hoopes C, Kukreja J, Golden J, et al. Extracorporeal membrane oxygenation as a bridge to pulmonary transplantation. J Thorac Cardiovasc Surg 2013; 145: 862-8.
15. Bermudez C, Rocha R, Zaldonis D, et al. Extracorporeal membrane oxygenation as a bridge to lung transplant: midterm outcomes. Ann Thorac Surg 2011; 92: 1226-32.
16. Fuehner T, Kuehn C, Hadem J, et al. Extracorporeal membrane oxygenation in awake patients as bridge to lung transplantation. Am J Respir Crit Care Med 2012; 185: 763-8.
17. Lang G, Kim D, Aigner C, et al. Awake extracorporeal membrane oxygenation bridging for pulmonary retransplantation provides comparable results to elective retransplantation. J Heart Lung Transplant 2014; 33(12): 1264-72.
18. Yusen R, Edwards L, Dipchand A, et al.; International Society for Heart and Lung Transplantation. The Registry of the International Society for Heart and Lung Transplantation: Thirty-third Adult Lung and Heart-Lung Transplant Report-2016; Focus Theme: Primary Diagnostic Indications for Transplant. J Heart Lung Transplant 2016; 35(10): 1170-1184.
19. Lehr C, Zaas D, Cheifetz I, Turner D. Ambulatory extracorporeal membrane oxygenation as a bridge to lung transplantation: walking while waiting. Chest 2015; 147(5): 1213-8.