Autor : EstĂ©vez-RamĂrez, Gloria M.1-2, MejĂa-Sandoval, Harvey J.3 , Saavedra, Alfredo4 , Callejas, Ana Milena5, Sánchez-Morales, Edgar A.4
1Fellow of Pneumology Universidad de la Sabana 2Internist physician Universidad Nacional de Colombia 3Endocrinologist Universidad Nacional de Colombia 4 Pulmonologist Universidad Nacional de Colombia - Instituto Nacional de cancerologĂa
https://doi.org/10.56538/ramr.NMDQ1943
Correspondencia : Gloria EstĂ©vez-RamĂrez. E-mail: gestevezr@unal.edu.co
ABSTRACT
Introduction: Thyroid cancer is the most prevalent endocrine neoplasm and the third
most common tumor in Colombian women. It has a good prognosis in the absence of
metastasis. The lungs are the most affected tissue by metastasis in patients
with thyroid cancer. The objective of this work was to describe the prevalence
of thoracic metastases in patients with thyroid cancer in a cancer reference
hospital from Bogotá, between 2016 and 2019.
Materials and methods: The study was cross-sectional, with non-probabilistic sampling of
consecutive cases in patients older than 18 with thyroid cancer who were
attended at the National Cancer Institute between 2016 and 2019.
Results: 241 patients were included in the study; 53 had thoracic metastases. The
mean age of patients with metastasis was 59.6 years (SD ± 12.7); 77.3% were
female and 90.5% showed papillary carcinoma. The prevalence of thoracic
metastases was 21.9%. The most common comorbidity was arterial hypertension
(32.08%). The most common symptom was dyspnea, with 15%. 34% of patients had
thyroglobulin of more than 38 ng/ml
and 22% had detectable anti-thyroglobulin antibodies.
Variables associated with
thoracic metastases were age (p = 0.002), weight (p = 0.019), cervical adenopathies (p = 0.007), presence of dyspnea (p = 0.004),
and thyroglobulin values greater than 38 ng/ml (p
< 0.001). Cervical adenopathies were found in 86%
of patients with metastasis, suggesting the fact that they are a risk marker
for metastasis.
Conclusions: This study allows us to establish local data, whose results promote
future prospective studies that identify associated variables and prognostic
factors in patients with thyroid cancer.
Key words: Thyroid neoplasms, Metastasis, Prevalence
RESUMEN
IntroducciĂłn:
El
cáncer de tiroides es la neoplasia endocrina más prevalente y el tercer tumor más
frecuente en las mujeres colombianas. Tiene buen pronĂłstico en ausencia de
metástasis. Los pulmones son el tejido más comprometido por metástasis en paÂcientes
con cáncer de tiroides. El objetivo del trabajo fue describir la prevalencia de
las metástasis torácicas en pacientes con cáncer de tiroides en un centro de
referencia en cáncer de la ciudad de Bogotá entre 2016 y 2019.
Material
y métodos: El
estudio fue de corte transversal con muestreo no probabilĂstico de casos
consecutivos en pacientes mayores de 18 años con cáncer de tiroides que
asistieron a consulta externa de del Instituto Nacional de CancerologĂa entre
2016 y 2019.
Resultados:
Se
incluyeron 241 pacientes; 53 presentaron metástasis torácicas. Dentro de los
pacientes con metástasis el promedio de edad fue de 59,6 años (DE ± 12,7),
77,3% fueron mujeres, y el 90,5% presentĂł carcinoma papilar. La prevalencia de
metástasis torácicas fue del 21,9%. Las comorbilidades más frecuentes fueron
hipertensiĂłn arterial (32,08%). El sĂntoma más frecuente fue la disnea en un
15%. El 34% presentaron tirogÂlobulina mayor de 38 ng/mL y un 22% tenĂan anticuerpos
antitiroglobulina detectables.
Las
variables asociadas a metástasis torácicas fueron la edad (p = 0,002),
el peso (p = 0,019), adenopatĂas cervicales (p = 0,007),
presencia de disnea (p = 0,004) y tiroglobulina
mayor de 38 ng/mL (p <
0,001). La presencia de adenopatĂas cervicales se encontrĂł en el 86% de los
pacientes con metástasis, lo que sugiere que se trata de un marcador de riesgo
de metástasis.
Conclusiones:
El
presente estudio permite establecer datos locales, cuyos resultados promueven
futuros estudios prospectivos que identifiquen variables asociadas y factores
pronósticos en pacientes con cáncer de tiroides.
Palabras
clave: Neoplasias
de la tiroides, Metástasis, Prevalencia
Received: 11/25/2022
Accepted: 08/16/2023
INTRODUCTION
Thyroid carcinoma is the most
common endocrine neoplasm;1 it has a low incidence, and it predomiÂnantly
affects the female population.2 In Colombia,
it is the third most frequent neoplasm in women3. Usually, it follows a benign clinical
course, espeÂcially when the histological type corresponds to differentiated
carcinoma (papillary and follicular carcinoma) and it has 10-year survival rates
that exceed 90%.3 However, the
presence of metastasis leads to a decreased life expectancy and a worse
clinical course of the disease.4
When there is metastatic
involvement, the lungs are the most commonly affected organs. The prevaÂlence
of lung metastasis from thyroid carcinoma varies in different cohorts and
ranges from 2% to 20%.5-7 Asian studies
have characterized lung metastases in patients with thyroid carcinoma. However,
in clinical practice, it is observed that not only pulmonary involvement occurs;
there is also mediastinal, pleural, intrathoracic airway, and chest wall involvement.8,9
The purpose of this study was to
determine the prevalence of thoracic metastases (lung, mediastiÂnal,
pleural, intrathoracic airway, and chest wall) in
patients with thyroid carcinoma, and to describe the sociodemographic,
histopathological, clinical, and exposure-related
characteristics of patients affected by thoracic metastases.
MATERIALS AND METHODS
An observational, cross-sectional
period study was conÂducted with retrospective data collection. The data were
extracted from the clinical records system of the National Cancer Institute
(INC). A sample size of 219 patients was calculated, aiming for 95% power and a
5% alpha error. Accounting for potential losses of 10%, a final sample size of
241 patients was determined. The study included patients with a confirmed
diagnosis of thyroid carcinoma, including all histological subtypes, who
attended the outpatient service between January 1, 2016, and December 31, 2019,
in Bogotá, Colombia. Individuals under 18 years of age, pregnant women, and
cases with more than 30% missing required data were excluded.
The independent variables
encompassed sociodemograÂphic factors, comorbidities,
previous exposures, treatment, thyroglobulin levels, anti-thyroglobulin
antibodies, and the primary dependent variable: the presence of thoracic meÂtastases.
Data collection was carried out using the Research Electronic Data Capture (REDCap) platform at the NCI.
A descriptive analysis was
performed, using measures of central tendency and dispersion (mean and standard
deviation, or median and interquartile ranges), depending on the distribution
of quantitative variables. Qualitative variables were expressed as absolute and
relative freÂquencies. Subsequently, a group comparison analysis was conducted
using statistical tests such as the Chi-square test or Fisher’s exact test,
based on the available data in each category for the collected variables,
mostly focusing on qualitative variables. Finally, a bivariate logistic
regression was conducted for the association analysis. The database was
analyzed using Stata 15.0 software.
Ethical considerations were duly
observed for the exeÂcution of this project, following both national and interÂnational
guidelines for clinical research involving human subjects. The researchers were
trained and certified in good clinical practices, and the project received
approval from the medical ethics committees of the Universidad Nacional de Colombia and the National Cancer Institute.
RESULTS
Between January 2016 and December
2019, 241 patients met the inclusion criteria. The primary outcome, the
prevalence of thoracic metastasis, was 21.9%. The identification of the
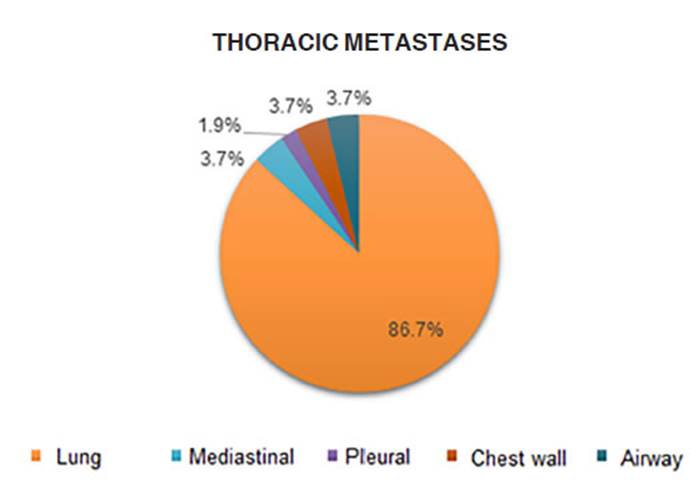
prevalence of each type of thoracic metastasis showed the following results:
19% for lung metastasis, 0.41% for pleural metastasis, 0.83% for mediastinal meÂtastasis, 0.83% for airway metastasis, and
0.83% for chest wall metastasis.
Among the total patients with
thoracic metastaÂsis, 86.79% of the cases were lung metastasis, 3.77% mediastinal metastasis, 1.9% pleural metastasis, 3.77% had
chest wall involvement, and in 3.77% of the cases, the airways were affected
(Figure 1).
77.35% of patients with thoracic
metastasis were female. The mean age of patients with metasÂtasis was 59.6
years (SD +14.3). 71.7% of patients were older than 55 years.
Regarding the histological type,
90.57% had papÂillary cancer, 1.89% had follicular cancer, 1.89% medullary
cancer, 1.89% anaplastic cancer, and 3.78% showed two histological types
(follicular and papillary, or follicular and medullary).
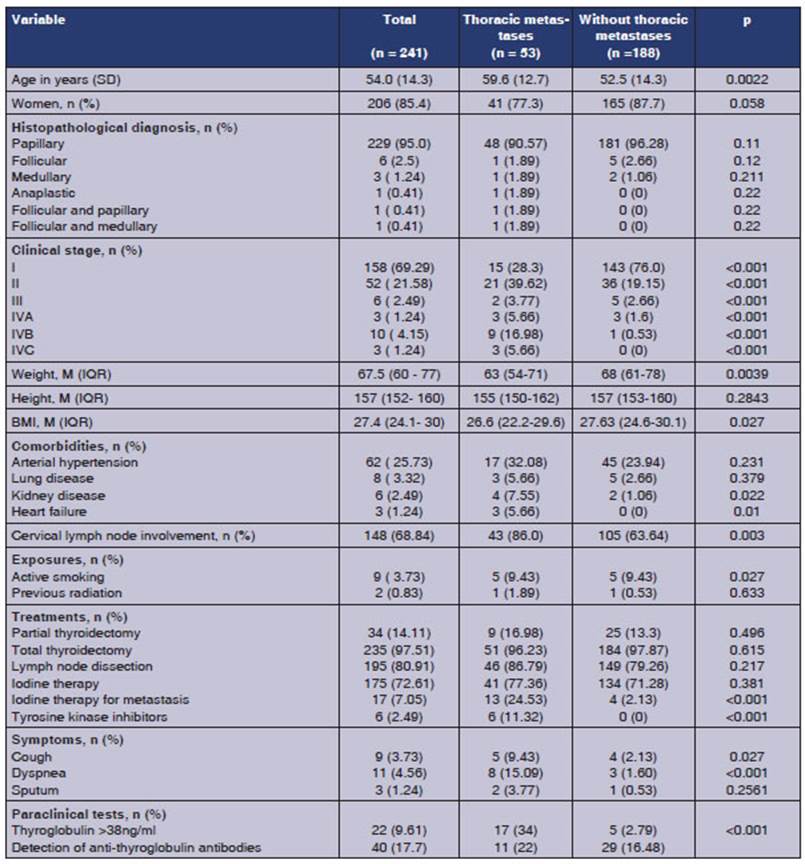
67.9% of patients with metastasis
showed stages I and II (annexes 1 and 2). The most common coÂmorbidities were
arterial hypertension (32.08%) and kidney disease (7.5%). 5.6% had associated
lung disease, and 9.43% and 1.89% had a history of active smoking and radiation
exposure, respecÂtively. 86% had concomitant involvement of cerviÂcal lymph nodes.
In terms of the treatments that were administered, 96.2% underwent total thyÂroidectomy,
86.7% lymph node dissection, 77.3% had initial iodine therapy, and 24.53%
required additional doses of radioactive iodine as part of metastasis
management. 11.32% of patients were managed with tyrosine kinase inhibitors.
Regarding the symptoms, 15.09%
presented with dyspnea, and 9.43% had cough.
When evaluating laboratory
results, it was obÂserved that 34% of patients with metastasis had a
thyroglobulin level above 38 ng/ml, which was
different from patients without metastasis, where such thyroglobulin values (above 38 ng/ml) were found in
2.7%. 22% of patients with thoracic metasÂtasis had detectable
anti-thyroglobulin antibodies.
When comparing patients with thoracic
metasÂtasis with those without metastatic involvement, a statistically
significant difference was found in variables such as: age (p=0.0022), clinical
stage (p = <0.001), BMI (p=0.027), kidney disease (p = 0.022), cervical
lymph node involvement (p = 0.003), active smoking (p=0.027), presence of
dyspnea (p=<0.001), and thyroglobulin above 38 ng/ml (p = <0.001).
Table 1 summarizes the
characteristics of the sample in general, as well as the characteristics of the
group with thoracic metastasis and the group without metastatic involvement.
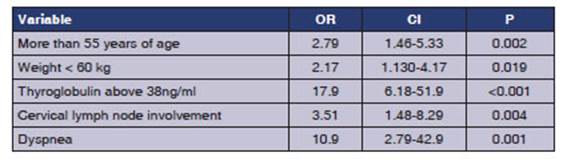
In the bivariate analysis, a
higher prevalence of thoracic metastasis was observed in patients over 55 years
of age, weighing less than 60 kg, with thyroglobulin levels above
38 ng/ml, and with involvement of cervical
lymph nodes and dyspnea. Results are shown in Table 2.
DISCUSSION
This study allows determining
local data regarding the prevalence of thoracic metastasis in thyroid cancer
and the factors associated with the presence of thoracic metastasis. This
information allows observing the frequency of this neoplasm when it shows
metastatic involvement and identifying variables that could be related to the
presence of metastasis. These would provide valuable inforÂmation for future
studies that contribute to the documentation of predictive factors for thoracic
metastasis in the local population with thyroid canÂcer. These data have not
been previously described.
The prevalence of thoracic
metastasis at 21.9% differs from national and international data. Data provided
by a study conducted in Manizales, CoÂlombia, reported an overall prevalence of
distant metastasis (both thoracic and extrathoracic)
of 1% in men and 1.04% in women10.
Regarding inÂternational data, the study of Chen et al. reported a prevalence
of 10.19%11.
The study by Song et al conducted in China, reported a prevalence of 7.95% for
lung metastasis. These percentage differences in prevalence could be attributed
to the fact that our population comes from a national cancer refÂerence center.
Sociodemographic data in terms of gender distribution align with the globally reported
preÂdominance of involvement in women2,12. 77.35% of
patients with metastasis were women, similar to the Asian cohorts of Sabra et al13 and Liete et al14,
who reported that 53% and 64.8% of patients with metastasis were women,
respectively. However, with regard to age, the mean age wasn’t similar to other
studies. In Brazil, a study conducted by Leiet et al14 found that
68.5% of patients were over 45 years old. In Asian studies like that of Sabrá et al13
and Chopra15,
patients with metastasis had a mean age of 52 and 45 years, respectively. This
suggests that Latin American populations likely tend to exhibit thoracic and
lung metastasis at a later stage compared to Asian populations. This finding
could be explained by genetic characterÂistics unique to our population or by
barriers of access to diagnostic methods within the system.
The distribution of histopathological types among patients with thoracic
metastasis was simiÂlar to what is reported in the global literature6,13,
with a higher prevalence of papillary cancer. ReÂgarding the stage at the time
of diagnosis, in this study, 67.9% of patients with metastasis were in stages I
and II at the time of diagnosis, contrary to a study in New York where 53% of
these patients were already in advanced stages (IV) at diagnosis, with only 38%
in stages I or II.13
A study conducted in the North
American popuÂlation16 reported
greater involvement of lung meÂtastasis along with the presence of cervical
lymph nodes in young patients, unlike the present study where 70% of patients
with thoracic metastasis and cervical lymph nodes were over 55 years old. This
difference in results suggests the hypothesis that in
the Latin American population, the behavior of the thyroid carcinoma generates
greater involveÂment in older individuals.
On the other hand,
the most reported symptom in our population with thoracic metastasis was
dyspnea. No studies were found in the literature that report
the symptoms of patients with metasÂtasis. However, a study conducted by Jang et al17 in patients who underwent iodine therapy
for lung metastasis reported that the presence of respiraÂtory symptoms was
associated with a decrease in forced vital capacity after the treatment. This
highlights the probable need for further studies in this area and the
development of a protocol for assessing lung function in patients who are to be
treated with radioactive iodine for thoracic and lung metastases.
In terms of follow-up
with laboratory studies, thyroglobulin and anti-thyroglobulin antibody tests
are of great importance. A Chinese study (Song et al) reported that 83.98% of
patients with lung metastasis had thyroglobulin levels above 100 ng/ml.6 In our study, 34% of
patients with thoracic metastasis and only 2.7% of patients without metastasis
had thyroglobulin levels of more than 38 ng/ml. This
value could be suggested as an estimated cutoff point for metastasis risk in
our population.
In our population, it
is a challenge to determine if the high prevalence and more frequent involveÂment
in older adults were a result of conducting the study in a cancer reference
center.
CONCLUSION
In conclusion,
considering the favorable prognosis of non-metastatic thyroid cancer patients
and the fact that pulmonary metastasis represents the most common form of
progression, it is imperaÂtive to continue documenting factors that could be
related to or associated with the presence of thoracic metastasis. These
factors could potentially serve as markers of specific and earlier intervenÂtions
in this population. The main strength of this study is the fact that it
provides local data on the prevalence of thoracic metastasis and potential
associated factors. This could prove valuable for future prospective studies
aimed at identifying associated variables and prognostic factors. The
disadvantage of this work is that it may be susÂceptible to classification and
information biases stemming from data loss, given the retrospective nature of
the study design.
Conflict of interest
The authors have no
conflict of interest to declare.
REFERENCES
1. Lebastchi AH, Callender GG. Thyroid cancer. Curr Probl Cancer. 2014;38:48-74. http://dx.doi.org/10.1016/j.currproÂblcancer.2014.04.001
2. Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA, JeÂmal A. Global cancer statistics
2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin.
2018;68:394-424.
https://gco.iarc.fr/today/data/factsheets/cancers/32-Thyroid-fact-sheet.pdf
3. Pardo C, Cendales R. Incidencia, mortalidad y prevalencia de
cáncer en Colombia, 2007-2011. v.1. Primera edición. Bogotá. D.C. Instituto
Nacional de CancerologĂa, 2015. p. 148.
4. Hernández A, Hurtado S, Silva L, Ortega C. CapĂtulo 37: Cáncer
de tiroides. En: Herrera-GĂłmez A, Ă‘amendys-Silva S,
Meneses-GarcĂa A editor. Manual de OncologĂa. 6e ed. MĂ©xico, D. f Mcgraw-hill Interamericana editores SA; 2017. p. 1–2.
5. Chala A, Franco H, Aguilar C, Cardona J. Estudio deÂscriptivo
de doce años de cáncer de tiroides, Manizales, Colombia. Rev Colomb Cir. 2010;25:276–89.
6. Song HJ, Qiu ZL, Shen CT, Wei WJ, Luo QY. Pulmonary metastases in differentiated thyroid
cancer: Efficacy of raÂdioiodine therapy and prognostic factors. Eur J Endocrinol. 2015;173:399-408. https://doi.org/10.1530/EJE-15-0296
7. Massin JP, Savoie JC, Garnier H, Guiraudon G, Leger FA,
Bacourt F. Pulmonary metastases in differentiated
thyroid carcinoma. Study of 58 cases with implications for the
primary tumor treatment. Cancer. 1984;53:982-92. https://doi.org/10.1002/1097-0142(19840215)53:4<982::AID-CNCR2820530427>3.0.CO;2-E
8. Dargent M, Colon J. Mediastinal
and pulmonary metastases from thyroid body cancers. JFORL J Fr
Otorhinolaryngol Audiophonol
Chir Maxillofac. 1972;21:583-4.
9. Dominiczak K, Fafrowicz B, Szmygin J. Thyroid cancer with metastases to the lungs. Gruzlica. 1968;36:1169.
10. Arias-Ortiz N, Guzmán-Gallego E. CaracterĂsticas clĂnicas del
cáncer de tiroides en Manizales, Colombia. 2008-2015. Rev
Peru Med Exp Salud Publica. 2020;37:287-91. https://doi.org/10.17843/rpmesp.2020.372.4892
11. Chen P, Feng HJ, Ouyang
W, Wu JQ, Wang J, Sun YG,
et al. Risk Factors for Nonremission
and Progression-Free SurÂvival after I-131 Therapy in Patients with Lung
Metastasis from Differentiated Thyroid Cancer: A Single-Institute,
Retrospective Analysis in Southern China. Endocr Pract. 2016;22:1048-56.
https://doi.org/10.4158/EP151139.OR
12. Haugen BR,
Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov
YE, et al. Thyroid Cancer: The American ThyÂroid Association Guidelines Task
Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid.
2016;26:1-133.
https://doi.org/10.1089/thy.2015.0020167
13. Sabra MM, Ghossein R, Tuttle RM. Time Course and PreÂdictors of
Structural Disease Progression in Pulmonary Metastases Arising from Follicular
Cell-Derived Thyroid Cancer. Thyroid. 2016;26:518-24. https://doi.org/10.1089/ thy.2015.0395
14. Leite AK, Kulcsar MA, De Godoi Cavalheiro B, et al. Death
related to pulmonary metastasis in patients with differenÂtiated thyroid
cancer. Endocr Pract. 2017;23:72-8. https://doi.org/10.4158/EP161431.OR
15. Chopra S, Garg A, Ballal S, Bal CS. Lung metastases from
differentiated thyroid carcinoma: Prognostic facÂtors related to remission and
disease-free survival. Clin Endocrinol.
2015;82:445-52. https://doi.org/10.1111/cen.12558
16. Showalter TN,
Siegel BA, Moley JF, Baranski
TJ, Grigsby PW. Prognostic factors in patients with
well-differentiated thyroid cancer presenting with pulmonary metastasis.
Cancer Biother Radiopharm.
2008;28:655–9. https://doi.org/10.1089/cbr.2008.0501
17. Jang EK, Kim WG,
Kim HC, et al. Changes in the pulmoÂnary function test after radioactive iodine
treatment in paÂtients with pulmonary metastases of differentiated thyroid
cancer. PLoS One. 2015;10:1–15.
https://doi.org/10.1371/journal.pone.0125114