Autor : Saraguro, Byron1, Torres, Otilia1, MenĂ©ndez, Denisse1, Rueda, MarĂa JosĂ©1, LĂłpez, MarĂa Fernanda1
1 Pulmonology Unit, Hospital General IESS (Ecuadorian Institute of Social Security), Babahoyo. Babahoyo-Ecuador
https://doi.org/10.56538/ramr.JLQN6996
Correspondencia : E-mail: byronsaraguromd@gmail.com (B. Saraguro)
CASE REPORT
24-year-old
male patient who is a university
student and practices mixed martial arts. He has no
personal history of pathological
conditions, but reports a surgery for an unspecified,
apparently benign mass on his
lower lip 15 years ago. He comes to the outpatient service due to a clinical condition characterized by cough and mild
hemoptysis of one month of evolution, dyspnea upon moderate
exertion that progresses to minimal exertion, and unquantified weight loss. No fever or night
sweats.
Physical examination: blood pressure, 100/70 mmHg; heart rate,
103 beats per minute; respiratory
rate, 16 breaths per
minute, with 95% oxygen saturation (FiO2 0.21).
On auscultation, diminished veÂsicular sounds were noted at the
left apex along with ipsilateral
pectoriloquy. Right lung field sounds
were normal.
Due to epidemiological history of residing in a region with high
incidence of tuberculosis, two
spuÂtum samples were taken for
bacilloscopy, both of which were negative.
The simple CT scan of the chest showed
decreased lung attenuation in the left field, predominantly
in the upper lobe and lingula, as well as a hypodense image in the right
pulmonary artery, indicative of pulmonary thromboembolism.
The contrast-enhanced CT
angiography of the chest showed absence
of the left pulmonary artery at the bifurcation level, and several collateral branches originating from a tortuous descending aorta with multiple ramifications
(Figures 1 and 2). Chronic thrombus
was also noted in the main
branch of the right pulmonary artery (Figure 3).
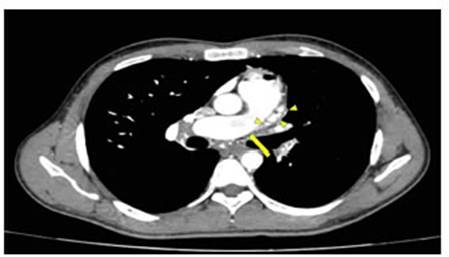
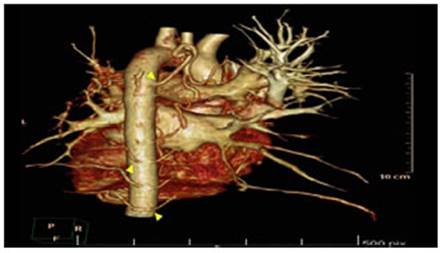
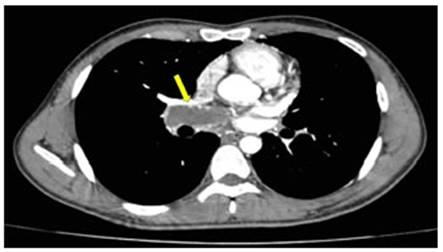
The electrocardiogram showed sinus rhythm,
pulmonary P wave, and signs
of right ventricle hyperÂtrophy and overload.
The transthoracic echocardiogram showed dilation of the right ventricle, dilation of the pulmonary trunk, and a systolic pulmonary artery pressure (sPAP) of 40 mmHg. No intracardiac shunts or masses were
observed.
Lower limb Doppler ultrasound was performed with
compressible vessels, without images inside, doppler flow present, negative
for deep vein thrombosis.
Laboratory studies reported normal blood count with platelets
at 179,000; D-dimer: 780; uric
acid: 7.4; non-reactive VDRL and HIV; PT: 16.6; INR:
1.53; PTT: 35.7; negative C-ANCA and P-ANCA; negative ANA; C3: 116; C4: 25; IgG
cardiolipin antibodies:
24.98; IgM cardiolipin antibodies: 12.27; rheumatoid
factor: 20.80; homocysteine 7.01; negative
direct Coombs test; negative
tumor markers; IgG and IgM for cytomegalovirus,
toxoplasma, rubella, herpes negative;
negative hepatitis C; CPK: 89; CKMB: 38.80; fibrinogen and coagulation factors within normal parameters.
It was concluded that the unilateral pulmonary artery agenesis in this patient was
not related to any cardiac anomalies,
and the pulmonary thromboembolism was idiopathic. The patient showed remission of hemoptysis, so anticoagulation with rivaroxaban was initiated.
Unilateral
agenesis of the pulmonary artery is a rare malformation
with a prevalence of one in every two
hundred to three hundred thousand young adults. Due
to its low frequency, it constitutes
a diagnostic challenge and is often underdiagnosed
in pediatric age.1
It occurs as a consequence of intrauterine involution of the ipsilateral sixth aortic arch, causing
a failure in the connection of this arch with the
pulmonary trunk, thus conditioning the absence of the proximal portion of the right or
left pulmonary artery.2
Pulmonary artery agenesis is more prevalent in the right artery, it
occurs in an isolated form, and is usually asymptomatic.
Agenesis of the left pulmonary artery is associated
with other cardiovascular malformations, such as tetralogy of Fallot, ventricular septal defect, aortic coarctation, pulmonary stenosis, and persistent arterial duct, which present more symptoms. When it is not
associated with these types of disorders, it is
considered an isolated finding.3
The most frequent mean age of diagnosis is around 14 years
old. It is
estimated that 13% to 30%
of patients may remain asymptomatic for many years,
and the disease is detected as a chance finding when performing
a chest X-ray.4
Patients may present with exertional
dyspnea, persistent cough, hemoptysis due to excessive aortoÂpulmonary collateral circulation from either hypertrophied bronchial collateral vessels or ipsilateral
peripheral arteriovenous
fistulas to the absent pulmonary artery (20% of cases), chest pain, respiratory
distress, heart failure, recurrent respiratory infections, or pulmonary hypertension
in 25% to 44% of cases.5
The physical examination is normal. Cardiac murmurs and pulmonary hypoventilation can be ausÂcultated, with or without pathological
sounds in the affected hemithorax.6
Definitive diagnosis is based on imaging
tests. The chest X-ray is
the first line of investigation, showing a size-reduced lung in the agenesis hemithorax
with ipsilateral mediastinal displacement, ipsilateral hemidiaphragm elevation, associated with contralateral pulmonary
hyperinflation.7 The
diagnosis is confirmed with contrast-enhanced tomography. The parenchyma may present mosaic atÂtenuation patterns, emphysematous changes, or bronchiectasis secondary to compensatory changes or recurrent
infections.8
The echocardiogram can
show associated cardiovascular malformations,
pulmonary hypertension, and
cardiac dextroposition. In most patients, collateral circulation develops from the
descending aorta, especially
the abdominal.9
The SPECT (single-photon
emission computed tomography) shows exclusion of
complete perfusion on the affected side,
with normal ventilation. Cardiac catheterization is necessary when
revascularizaÂtion is
planned.10
The multiphase MR (magnetic resonance) angiography allows for obtaining anatomical
and funcÂtional information
of thoracic vascular structures
with a single injection of intravenous contrast and without the use of ionizing radiation.11
Lung function tests show normal or restrictive pattern with normal diffusion capacity.
The electrocardiogram is usually normal but may show right
ventricular dominance in cases associÂated
with pulmonary hypertension12
Conservative treatment
is indicated for asymptomatic forms, and surgical treatment for cases with symptoms or
serious complications.
Surgical management by lobectomy or
pneumonectomy with selective embolization is performed in patients with massive
hemoptysis or recurrent lung infections. Repair surgery consists of two stages: surgical
anastomosis of the proximal and distal parts of the pulmonary
artery, followed by stent imÂplantation.13
Approximately 19% to 25% of patients with congenital
absence of the pulmonary artery present with pulmonary
hypertension later in life, which implies
a poor prognosis, with a mortality rate of approximately 7%. This can be treated with antihypertensive
drugs.
Death can be secondary to massive hemoptysis, respiratory failure, pulmonary hypertension leading to right heart failure.
No
cases of pulmonary thromboembolism
associated with unilateral absence of the pulmonary artery have been described;
however, chronic thromboembolic pulmonary hypertension has an incidence of 0.01% to 9.1% after an acute pulmonary
embolism and can be resolved by
thromboendarterectomy.14
Volkan et al. reported
a case of left pulmonary artery agenesis without congenital cardiac anomaÂlies, similar to the patient described
in this review, which could contribute
to the maintenance of an asymptomatic state.
The diagnosis of unilateral agenesis
of the pulmonary artery can be considered a challenge, and a high level of suspicion would help obtain
a proper diagnosis.15
Conflicts of interest
The authors have no conflict of interest to declare.
REFERENCES
1.
Adán V, Jiménez A, Martín C, García J. Agenesia
aislada de la arteria pulmonar derecha. An Pediatr. 2017;86:45-49. https://doi.org/10.1016/j.anpedi.2016.04.003
2.
Serra W, Tafuni F, Sverzellati
N, Cattabiani M. Pulmonary Artery Agenesis in Young Adult – Two Case Reports. Int J Clin Cardiol. 2017,4:096.
https://doi.org/10.23937/2378-2951/1410096
3.
Cáceres González JD, Cáceres Acosta MF, Osorno Serna J,
Gómez Correa GA, Rodríguez Reyes FA, Suarez Poveda T. Agenesia de
la arteria pulmonar izquierda: reporte de caso. Rev Colomb Neumol. [Internet].
2021;33. https://revistas.asoneumocito.org/index.php/rcneumologia/article/view/521
4.
Darwazah AK, Alhaddad IA. Pulmonary artery agenesis associated with coronary collaterals
among adults. J Cardiothorac Surg. 2016;11:109. https://doi.org/10.1186/s13019-016-0504-1
5.
Ashiq Zindha B, Oommen BE, James BC, Joshi A, Dhake S, Jethwani J. CT features of unilateral agenesis
of pulmonary artery. Eurorad https://doi.org/10.1594/EURORAD/CASE.11675
6.
Narra RK, Annareddy M, Janam
R, Syed S. Unilateral agenesis
of the pulmonary artery (UAPA) in an adult. BMJ Case Rep. 2022;15:e248397.
https://doi.org/10.1136/bcr-2021-248397
7.
Moosavi SA, Iranpour A.
Unilateral pulmonary artery
agenesis in an adult patient with
cough and hemoptysis: a
case report. Tanaffos. 2014;13:58-60.
8.
Maggiolo J, Rubilar L.
Agenesia unilateral de la arteria pulmonar. Presentación de dos casos y
revisión de la literatura. Neumol Pediatr. 2021;16:48-52.
https://doi.org/10.51451/np.v16i1.236
9.
Saladi L, Roy S, Diaz-Fuentes
G. Unilateral pulmonary artery
agenesis: An unusual cause of unilateral ARDS. Respir
Med Case Rep. 2018;23:148-51.
https://doi.org/10.1016/j.rmcr.2018.02.004
10.
Pla A, Pineda V, Roche S. Agenesia unilateral arterial pulmonar, hallazgos por angio-RM. Rev Esp
Cardiol. 2013;66:821.
https://doi.org/10.1016/j.recesp.2011.12.012
11.
Rodríguez-Gómez F, Martín I, Sánchez A, Pujol E.
Edema de pulmón unilateral e hipertensión pulmonar tratada con sildeÂnafilo en la agenesia de la arteria pulmonar. Rev Esp Cardiol.
2006;59:1345-50. https://doi.org/10.1157/13096595
12.
Farghly E, Bousamra M. Hemoptysis resulting from unilateral pulmonary artery agenesis. Ann Thorac Surg. 2002;74:255-7.
https://doi.org/10.1016/S0003-4975(02)03558-0
13.
Johnson TR, Thieme SF, Deutsch
MA, et al. Images in cardiovascular medicine:
unilateral pulmonary artery
agenesis: noninvasive diagnosis
with dual-source computed tomography. Circulation. 2009;119:1158-60.
https://doi.org/10.1161/CIRÂCULATIONAHA.108.777698
14.
Hon S, Channick RN, Farber HW. Unilateral Chronic Thromboembolic Pulmonary Disease: A Mimic of Pulmonary Artery Agenesis. Am J Respir Crit Care Med.
2020;201. https://doi.org/10.1164/rccm.201905-0997im
15.
Emren SV, Tülüce
SY, Tülüce K. Isolated
Congenital Unilateral Agenesis
of the Left Pulmonary Artery with Left Lung
Hypoplasia in an Asymptomatic Adult Patient. Acta Cardiol Sin. 2015;31:572-5. https://doi.org/10.6515/ACS20150511B