Autor : Rey, DarĂo R.1
1Consultant physician in Pulmonology, Hospital Gral. de Agudos Dr. E. TornĂş
https://doi.org/10.56538/ramr.CVPR8593
Correspondencia : DarĂo R. Rey E-mail: darioraul.rey@gmail.com
OCCUP DILD SIMILAR TO CHRONIC ORGANIZING PNEUMONIA (COP)
COP is defined by a histological pattern,
and the corresponding clinical, raÂdiological, and pathological diagnosis is
cryptogenic organizing pneumonia when there isn’t a clear cause.
Due to the fact that there may be
outbreaks in the distal bronchioles, the disease was previously called
“bronchitis obliterans with organizing pneuÂmonia”
(BOOP); that was the typical, predominant pathological pattern. At present, it
is internationally recognized as COP.1
After assessing the clinical,
tomographic, and histopathological features that lead
to the COP diagnosis, other disorders have to be considered, such as tumors,
infectious processes or inflammatory coditions of the
lung. AlÂthough the histological pattern of COP is non-specific, it can be
related to other diseases, thus it has a relative value when it is found in a
sample of such size. The first step for a presumptive diagnosis can be the
chest CT (computed tomography). When COP shows unique or multiple areas of paÂrenchymal
consolidation, the differential diagnosis includes lepidic
carcinoma, pulmonary eosinophilia, Churg-Strauss
syndrome, rheumatoid arthritis, polymyositis,
radiation therapy or the consequence of the use of drugs or monoclonal
antibodies.
In relation to the Covid 19 pandemic, there are COP publications as a result
of the subacute evolution of this new viral disease.2-10
Zhang et al reviewed 1,346 cases
of COP from the Shanghai Pulmonary Hospital in the period between January 2000
and December 2000. The cause was diagnosed in 1,170 patients (86.9%), whereas
in 176 cases (13.1%) the origin was unknown. Only in 13 cases, the disease was
related to the indiÂvidual’s work, including 2 welders, 3 assembly line workers
and 3 textile machine operators. 4 foundry workers and 1 case with prolonged
exposure to glass dust.11
GIANT CELL INTERSTITIAL PNEUMONIA (GIP)
GIP is a serious and rare
occupational lung disease that occurs as a consequence of exposure and asÂpiration
of hard metals with abrasive properties (tungsten carbide and cobalt). Most of the
time, exposure occurs in the industry of cemented tungÂsten carbide, mining
processes, manufacturing of alloys, and polishing and grinding of tools with
the grinding wheels of hard metals. People with chronic exposure can develop
interstitial lung disÂease and show worsening of dyspnea, nonproducÂtive and
persistent cough and exercise intolerance.
In the initial classification of Liebow and CarÂrington it appeared as a disease of the interstitium, but then, once its relationship with hard
metals had been proven, it was reclassified as pneumoÂconiosis, also after it
was widely recognized that GIP is characterized by a histologic pathognomonic
pattern of multinucleated cells.12,
13
Cobalt has several industrial
uses but not all of them cause OCCUP DILDs (occupational diffuse interstitial
lung diseases). It occurs when workers are exposed to cobalt through the
manufacture or use of tools created for the process of powder metallurgy. This
is a procedure for the manufacÂture of metal objects. It starts from fine dusts
that are then compacted and are finally given a certain shape by heating them
at a determined temperature to obtain a tool. When tungsten and cobalt are
heated, they combine to form a tight metal matrix.
The worker who is specialized in
powder metalÂlurgy is an expert in such techniques. The manuÂfactured pieces
show special characteristics, such as lubrication or anti-friction. In the
industry of diamond tools, cobalt dust is used as a matrix for diamonds.14 One patient revealed he had worked as a plumber in the oil
industry, from 1982 to 1991. During that period, he frequently assembled pipes
using sanders and grinders with hard metal discs, which he grinded every time
they went blunt. When he did that, he only wore a visor. When the symptoms began,
an X-ray was performed, showing a lung infiltrate.15
On other occasions, as in the
description of Carmo-Moreira, the symptoms began with
a sponÂtaneous pneumothorax in a worker whose job was to sharpen saws and
knives, and he had done that job for 8 years without protection.16
The consulted literature includes
some obserÂvations; some of them draw our attention due to the number of cases
being described. Between 1985 and 2016, Chiarchiaro
et al identified 23 patients with a pathological pattern, 93% of which showed
“ground glass”, and 85% of those showed GIP in the biopsy that was performed.
Thanks to corticosteroid treatment, these cases had a better evolution.17
Naqvi et al reviewed 100 cases of these pneuÂmoconiosis
that had been studied for 50 years. GIP was histologically proven in 59 cases,
and in the remaining 41 it was confirmed through scanÂning electronic
microscopy and x ray spectroscopy. The cases of GIP in the industry of cemented
tungsten carbide revealed high concentrations of tungsten, though cobalt was
only detected in 6% of the cases.18
To conclude, as a
non-work-related etiology, there are publications that relate GIP with the
prolonged use of nitrofurantoin.19,
20
HYPERSENSITIVITY PNEUMONITIS (HP), AN OCCUP DILD:
HP is produced immunologically by
the repeated inhalation of a great variety of chemical substances or
environmental organic antigens to which a genetically susceptible individual
has been previÂously sensitized.
Alternative definitions for HP
have been proÂposed, but experts disagree on how to describe the disease in
detail and use their diagnostic orientaÂtion and criteria.
Maybe one of the most complete
definitions is the one of Cormier, who defines HP as “An inadequate immune
response to inhaled antigens that causes difficulty to breathe, a restrictive
pulÂmonary defect, and interstitial infiltrates observed in lung images (chest
X-ray and high resolution tomography) caused by the accumulation of a high
number of T lymphocytes activated in the lungs. On some occasions, the
disease is also characterized by fever episodes a few hours after the exposure”.21, 22
According to Hirscmann
et al, from the tomoÂgraphic viewpoint, HP can be classified as ACUTE, when it
shows “ground glass”, centrinodular pattern and air
trapping, and CHRONIC, when there is reticulation, “honeycombing”, peribronÂchovascular thickening and lung architectural
dislocation.23
Therefore, the possibilities of
suffering HP, whether it is occupational or non-work related, are unlimited, and
there may exist as many observaÂtions and/or publications as potential
offensive antigens. One proof of that is found in the ConsenÂsus published in
2020 by the American Thoracic Society, the Japanese Respiratory Society and the
Latin American Thoracic Association.
Table 1 shown in the article
includes sources of known antigens that cause HP, for example: organic
particles, yeast, environmental fungi, protozoa, bacteria, enzymes, animal and
plant proteins, inorganic substances, pharmacological agents, and metals.24
Yoshida et al conducted a
national investigation to look into the epidemiological and clinical characÂteristics
of HP in Japan. 185 doctors completed a questionnaire, and 835 cases were
classified as HP, with occupational HP in 115 of those cases (13.8%), and
predominance of “farmer’s lung disease” (59%). 19 of the workers manipulated isocyanates and 10 office clerks had suffered
microbiological contamination through the air conditioner. The investigation
concludes with the recommendation of a thorough environmental assessment and a
panel of antigens adapted to exposure variations, as diagnostic orientation.25
Moon Bang et al reported that
there are few studies in the United States population that invesÂtigate HP.
National data of the NIOSH (National Institute of Occupational Safety and
Health) can contribute to understand the epidemiology of this disease. They
analyzed the identification of causes of death for the 1980-2002 period. Mortality rates according to the type of industry
and occupation were adjusted per age, sex and race in 26 states that provided
industry and occupation informaÂtion between 1985 and 1999. This mortality rate
for HP was significantly high for the agricultural and livestock production,
also for farmers. They conclude their investigation by saying that agriÂcultural
industries are closely associated with HP mortality, thus it is extremely
important to evaluÂate preventive strategies to protect the workers of these
industries.26
The literature review allows us
to appreciate that there is an overlapping of publications about occupational
asthma (OA) and the OCCUP DILD HP. When evaluating both, it is estimated that
in sensitized individuals, progression from one disease to the other would
depend both on the conÂcentration of the offensive agent and the prolonged
period of exposure with inadequate protection or without any protection at all.
Like the expert Salvaggio said in an editorial 50 years ago, HPs (occupational
or non-occupational) are a kind of “Pandora’s box”, and research could provide
the suitable answers for each particular case.27
The HPs more frequently cited in
the literaÂture are: “farmer’s lung disease”, “baker’s lung disease”, the ones caused by MWF (metalworkÂing fluids) and
isocyanates and those caused by waterproofing
substances.
“Farmer’s lung disease”
An
eventually serious disease that results from the manipulation of moldy and
dusty organic materiÂals. Exposure
to such materials induces the disease in certain people, whereas other people
are not affected. The lung biopsy reveals granulomatous interstitial
pneumonitis. Individual hypersensitivÂity to fungi or fungal products seems to
be a crucial factor in getting this disease. The treatment is to avoid exposure
of sensitized workers. The thermoÂphilic actinomycetes, Saccaromyces rectivirgula, Termopolyspora polyspora or Micropolyspora faenii are considered particularly important anÂtigens
responsible for most reported cases of HP. Campbell was the first to consider
causality, even though he didn’t call it that way, and Fawcitt
thought it was related to aged, moldy cereal. 28, 29
An epidemiological
survey conducted among farmers in China showed 6% of producers diagÂnosed with
HP: 19% of them had occupational asthma, and 17% had COPD (chronic obstructive
pulmonary disease). Influencing factors were high humidity and the high
capacity and short height of warehouses.30 Factors contributing to
agricultural HP in France were high humidity and tight hay packing, correlated
with a higher concentration of HP-promoting microorganisms. As a prevenÂtion
measure, respiratory protection shall be used when packing down the hay and
manipulating potentially moldy hay and during forage drying.31
Over the decades,
numerous studies have been reported on this condition and its relationship with
rural tasks, with Peppys’ work being essential for
its research and serological confirmation after studying 327 agricultural
workers with different antigens, where 89% showed positive reactions; and 87%
of 205 farmers were due to hay contamiÂnated with Termopolyspora
polyspora.
The higher the
serological titers, the more reactions there were to other antigens,
and the more severe and frequent the episodes, with a male predominance. Only
18% of 122 non-exposed farmers who didn’t have the disease showed reactivity.
Peppy concludes by emphasizing that the “farmer’s lung” disease was insidious
in 49%, sub-clinical in 9% and with typical symptoms in 32%.32-34
This occupational
disease is not usually seen in our environment because the cattle are fed on
pastures rather than stored hay or cereal that favor
fungal contamination.
Cuthbert and Gordon
conducted a 10-year follow-up study of 29 cases of this disease. The results
revealed that respiratory protection and the replacement of hay with pasture
stored in silos favored prevention. To be safe, respirators should be used in
situations where there is agricultural dust, especially in enclosed environments.35
In recent decades,
the frequency with which chronic bronchitis, non-smoking-related emÂphysema,
and tomographic signs of HP appear in this disease has been reported. Depierre et al investigated 1,763 rural workers in France
with serology and questionnaires, obtaining a response in 69%, out of which 270
were suspected of having the disease. They found a relationship between chronic
bronchitis and this condition, and sugÂgested that fungal dust was responsible
(50.6% in those affected versus 8.6% in controls with a p < 001). They
concluded that chronic bronchitis in farmer’s lung was independent of smoking
and age. There were 9.2% radiological abnormalities of the lung interstitium and were less common in plains or mountains,
probably due to cold enviÂronmental conditions.36 As
a rare case, Soumgane et al describe a woman with
“farmer’s lung” who showed PEEP (positive end-expiratory pressure). She had
excellent evolution with corticosteroids and 1-year follow-up.37
Lung interstitium disease
due to metalworking fluids (MWF)
The MWFs are
essentially oil-in-water emulsions with additives (corrosion inhibitors,
emulsifiers, anti-foaming agents, and biocides). Their microÂbial contamination
is almost systematic, as their components serve as nutrients for contaminating
microorganisms. Biocides for MWFs are protecÂtive products used to counteract
microbial conÂtamination and growth. (The appropriate criteria for a biocide
for MWFs are: 1. Broad-spectrum activity. 2. Suitable for low concentrations.
3. Compatible with the formula and physicochemical properties of the MWF and
stable over time. 4. Effective in the presence of dirt. 5. Non-corrosive to
metals. 6. Safe for people and the environment. 7. Economical.
The future lies in
developing new molecules with biocidal activity that
correspond to: A.- Optimizing the performance of
current molecules. B.-Establishing different strategies to
enhance biocidal activity. With over 1.2
million workers in the US involved in the manufacturing of machinÂery, machine
tools, and automobiles, exposure to MWFs is common.8, 39
Epidemiological
surveillance methods are useful for revealing causality by demonstrating that
the MWFs are the most common factors in occupational asthma, along with isocyanates. In Bakerly’s
publication, they accounted for 11%, and the latter for 21%, while in the
publication of Rosenman et al, MWFs accounted for 11%
and isocyanates, 14%.40, 41
There are periodical
publications of cases or series of cases of occupational asthma caused by MWFs,
whereas those of HP caused by the same products are less frequent.42-45
In 1995, Bernstein et
al published the first 6 cases of HP caused by MWF, and episodes of this
occupational disease appear relatively freÂquently. 46-49
Systematic studies
show that both HP and ocÂcupational asthma are caused by fluids, but also by
microorganisms or fungi that grow in them, mainly Mycobacterium immunogenum, which is responsible for contamination and
causing hyperÂsensitivity in experimental animals.50-52
“Baker’s lung” disease
Baker’s asthma is one
of the most common causes of occupational asthma, and its incidence is estiÂmated
to be between 1-10/1,000 bakery workers. A bakery establishment is a complex habitat
with an unlimited number of potential sensitizers. EmÂployees in this industry,
including millers, bakers, and food processors who are exposed to bakery
allergens, may develop this disease. The main alÂlergens are the flour (wheat,
rye, and barley), the enzymes added to the dough (such as α-amylase), and the parasites and fungi that can
contaminate the flour. This type of occupational asthma is IgE-mediated
(mediated by immunoglobulin E); and titration of IgE
is essential for the diagnosis of the condition.53, 54
A study published by Simonis et al studied in 433 bakers the IgE
and IgE levels specific to baking enzymes
investigated in the Asthma Prevention Program at the German Social Accident
Insurance Institution for the Woodworking and MetalworkÂing Industries,
calculating personnel exposure to environmental dust, including the
concentration of the α-amylase level in the
work area.
They reached the
following results and concluÂsions:
a. Significant
decrease (from 26% to 13%) in senÂsitization to α-amylase.
b. Sensitization to glucoamylase was much higher than to cellulase.
c. Sensitization to
all three enzymes is common in bakers.
d. 30% of bakers are sensitized to at least one of the
enzymes.
e. Exposure to α-amylase has decreased.
f. 11% fewer bakers are exposed to α-amylase, compared to 10 years ago.
g. The high
sensitization to glucoamylase in afÂfected bakers
leads to investigating exposure levels in bakeries and evaluating
sensitizations in the context of occupational diseases.55
The research of Diederichs and Lubers from 60
years ago revealed a sensitivity incidence of around 54 percent among bakers.
The expected presentaÂtion of signs and symptoms was estimated at 12.7 years
for occupational rhinitis and 15.3 years for occupational asthma.56 In 1980, Thiel and Ulmer published a comprehensive study
reporting that in Germany it was a recognized occupational disÂease. In ancient
Rome, it was known that slaves who made bread had great suffering, and the
first scientific reference was due to Ramazzini
around 1700. However, HP caused by flour, parasites, fungi, or enzymes is
exceptional. The publication by Gerfaud et al on HP
in a baker showed that there was good evolution with corticosteroids and mycophenolate, but the serology was positive for corn,
oats, Aspergillus fumigatus,
and mites such as Glyciphagus destructor or Sitophilus granarius, thus
showing the complexity of the diagnostic studies required when testing, in this
case, 26 antigens.57, 58
A case of HP caused
by flour parasites has been published, but the most interesting one is that of
van Heemst et al about HP induced by phytase in a worker who performed his tasks for 20 years
without protection, producing food for chickens. Phytase
catalyzes the hydrolysis of phytate, which is a way
of storing the phosphate existing in soy and cereals. Poultry and pigs use this
phosphorus partially, so in order to increase its availability, the enzyme is
added to the food.59, 60
To conclude, Brant et
al conducted a survey and performed serological tests in 239 bakers from
different British supermarkets. Results showed that 15% had respiratory signs
and symptoms, 11% had positive serology for flour, and 4% for α-amylase. Despite their low levels of dust expoÂsure,
this population of bakers shows significant levels of sensitization and
respiratory symptoms related to their work. Changes in the workplace and
modifications in the bread-making process have caused a shift in the
distribution of occuÂpational asthma and HP among bakers in the United Kingdom.61
“Bird fancier’s lung” disease
The bird breeder is
exposed to an immunological lung disease due to repeated exposure to avian
antigens transmitted through the air. It is a type of HP triggered by the
excretion of highly antigenic avian proteins and/or waxy proteins that cover
the feathers of a variety of birds, causing a hypersenÂsitivity reaction in a
susceptible host.62
The disease may be
more a consequence of a recreational activity than a work-related one. This
condition, which in the vast majority of cases is expressed as occupational
asthma, is associated with a variety of abnormal findings: skin tests,
radiographic abnormalities, serology, and disturÂbances of the lung function.
Unfortunately, none of these are diagnostic, the
disease is best identified through clinical criteria.63
To that end, Morel et
al studied 86 patients with HP between 1977 and 2003, where one-fifth of the
patients had the chronic form of the disease. All of them were studied with
serology, chest radiograÂphy and CT, skin tests, FBC (fibrobronchoscopy)
with BAL (bronchoalveolar lavage) and/or TBB (transbronchial biopsy). 82% had cough, and 98% had dyspnea,
with 25% in functional class III or IV and 18% with chest tightness.
Lymphocytosis was found in 83% of BALs, and the CT showed 79% of interstitial
pattern and 68% of ground glass opacÂity. Serology was positive in 92% of the
series. 64
Serology is of great
value in collaborating with the diagnostic suspicion. The ELISA method (enÂzyme-linked
immunosorbent assay) used proved to be useful for
evaluating specific IgG responses. In a meta-analysis
carried out by Shiroshita et al, ELISA showed high
sensitivity, and the OuchterÂlony method exhibited
high specificity.65, 66
The study of McSharry in 50 affected individuals to validate an
automated fluorometric antibody detection procedure
provides a method for interÂnational standardization of HP, thus improving
quality control and refining its suitability as a diagnostic complement.67
There are relatively
few publications about HP caused by bird antigens, most related to pigeon
activity, but it is worth noting the case of Chopra, with exposure to birds for
35 years, or that of Cooper, in which the person’s job was cleaning in a
restaurant and collecting duck and goose feathers which he then placed in a
vase in his home for the last 6 months. Outside the individual’s home, there
were no birds. Sometimes, it is presented with unexÂplained dyspnea or as an
expression of a COP.62, 68, 69
Induction by isocyanates
In many nations, isocyanates (ICN) are a very common cause of occupational
asthma. Although this reference is very important and HP has been occasionally
reported, it may be a more common result than originally believed as a
consequence to ICN exposure.
ICNs are used in the
manufacturing of a wide variety of products, especially in the production of
flexible urethane foam, lacquers, varnishes, paints, and rubber modifiers.
Their toxicity has been known for decades: cited by Blake et al, the first
description was in 1951 by Fuchs; and Schurman and
Rein reported two cases of patients who died from severe asthma in 1955.70
Little is known about
the inevitable occupaÂtional levels related to the induction of HP by ICN. 60
By performing adequate
environmental monitorÂing and strict medical-occupational control, expoÂsure to
ICN is associated with low sensitization and minimal exposure to the causative
agent. 71 Unprotected exposure can cause dermatitis, conjunctivitis,
rhinitis, “industrial” bronchitis, occupational asthma (which is the most
common finding in the literature) or, every now and then, HP. 72 Baur reported 14 cases of HP caused by ICNs when
investigating 1,780 workers who used this material, , representing 1% of the
workforce, while Vandenplas found 4.7% in his
research. The difference would result from different working conditions.73,
74
Particular susceptibility plays
an important role, as in the case of HP caused by ICNs in a company secretary
who went several times a day to the premises where this causative agent was
being used to dye boots.75 Treatment with steroids can give
excellent results, and in Japanese literaÂture, there is a publication of an
individual who got HP caused by ICNs while painting a car as a recreational
activity.76, 77
Permitted environmental limits of
ICN in the US have been decreasing from 0.1 ppm in 1956 to 0.005 ppm/8 hours of
work or 0.02 ppm for 4 periods of 15 minutes/day in 1980.78
Minimizing or preventing exposure is essential in occupational medicine, health
and safety. It is essential to conduct educational talks; and medical superviÂsion
must be carried out by performing periodic spirometries
to test the workers so as to detect functional changes. In areas with higher
concenÂtrations of ICNs, Nakashima et al recommend performing specific IgE serological controls, too, to enable early detection
and take appropriate action.79
HP by waterproofing agents
Waterproofing agents are used to
coat leather, fabric, or solid surfaces in order to ensure resisÂtance to dirt
and water. They typically consist of 3 components: an active compound (water
repellent), a solvent, and a propellant (propane, butane), if they come in a
can. The water repellent is a mixÂture of siloxanes
or acrylate polymers that contain fluorocarbon or hydrofluorocarbon.
Nowadays, aqueous mixtures of glycols and glycol ethers are often used as
solvents.80
Over the past 20 years, different
health effects from the use of waterproofing agents have been described in
approximately 20 reports involving the exposure of more than 200 people.
Isolated cases related to
waterproofing agents often appear, and in the majority of cases, volatile
organic compounds (VOCs) play an important role.81-83
Scheepers et al published the impact on 10 workÂers exposed to a waterproofing
agent with a low percentage of VOCs and nanoparticles. To sum up, a worker who
had smoked right before entering the workplace was hospitalized with injuries
in both lungs, and the other nine experienced respiratory symptoms within 24
hours of entering the work environment.
After the relevant studies had
been conducted, the authors concluded that the hospitalized workÂer’s
cigarettes were contaminated with the liquid. The symptoms of the other workers
were due to suspended material still present in the environÂment. The volatile
compounds could have been at play if the building was completely enclosed.84
A very interesting observation is
that of Tan et al, who published 11 cases, with 5 cases of respiraÂtory
distress and 1 death. The research revealed that a neighboring factory, 35
meters away from the affected workshop, had released fluorocarbon waste without
spraying water on the waste, causÂing the inevitable accident.85
In Switzerland, between October
2002 and March 2003, there was an acute outbreak following exposure to
waterproofing agents. 180 cases were reported (previously, less than 10 cases
per year had been registered). The reported cases involved 3 brands of aerosols
that had changed their forÂmula prior to the incident. A retrospective analysis
was carried out to clarify the circumstances and causes of the observed
effects. The results obtained showed high variability of individual responses,
suggesting that some indirect mechanism predominates in the incidence of the
disease. The findings suggest that improvements in environmental exposure
conditions are not sufficient to prevent future toxic outbreaks due to
waterproofing spray. More effective preventive measures are suggested to be
taken before marketing and distributing new waterproofing products.86
Repeated unprotected exposure to
the causative agent for 4 years can lead to the evolution of chronic HP, as
described in a publication from 2017, considÂered as the first known case at
that time. 87
OCCUP DILD induced by green tea
During the production of green
tea, a fine powder called “tea fluff” is released into the factory’s
atmosphere. Inhalation of this powder can cause respiratory distress relatively
quickly. Chronic cough in tea factories and tea taster’s disease are two
occupational diseases associated with the industry of this product.88
The first case to be published was about occupational asthma caused by tea
dust, and was confirmed by intradermal skin testing and specific bronchial
challenge, although this author cites observations by Castellani
in Ceylon dating back to 1919.
Cartier and Malo
reported on 3 similar cases studied by them, and the Japanese literature
highlights publications of HP caused by green tea, with the publication of
Tanaka et al being notable for the high environmental concentration of the
causative agent in the factory premises.89-93
Green tea has 8% epigallocatechin compared to black tea, which has 1%, and
is the main cause of occupational asthma and HP. Shirai
et al observed a significant correlation between the maximum percentage of
histamine release and epigallocatÂechin concentration
in specific intradermal reacÂtions, as well as positive results when doing the
bronchial challenge test with green tea dust.94, 95
Miscellaneous
As previously explained, the
inhalation of organic particles (animal proteins, fungi, or bacteria) or
workplace materials can induce the appearance of HP in sensitized individuals.
The literature is constantly updated with cases related to unexÂpected antigens
and individual susceptibility, so it is impossible to dominate it in full.
Interesting observations can be
gleaned from it, such as those of cork workers (Suberosis),
“cheese washers” where the responsible antigen is Penicillium
cassei or Roquefortii, HPs
caused by air conditioning contamination, and those in mushroom processing, or
an exceptional case, such as that of Marchisio et al,
caused by the contamiÂnation of deli meat with Penicillium
camembertii in a sausage factory. 96-108
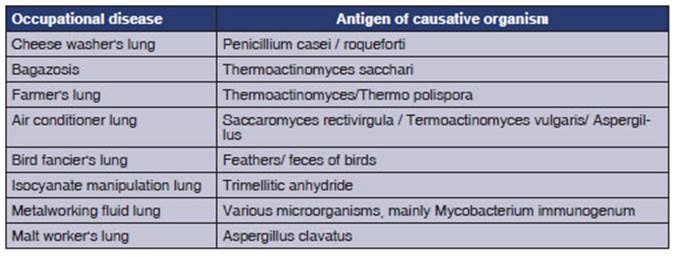
To conclude, we attach a concise
table containÂing some occupational lung diseases that can lead to HP and their
common causes. (TABLE)
EMERGING OCCUP DILD (NEW WORK-RELATED DISEASE?)
Cummings et al published a
comprehensive study conducted in production areas of an industrial machinery
factory, where five previously healthy non-smoking men who worked between 1995
and 2012 developed respiratory symptoms.
They all presented with a gradual
onset of cough, sibilance, and dyspnea on exertion with an average decrease of
44% in predicted FEV1 (forced expiratory volume in the first second) and 53% in
DLCO (diffusing capacity of the lungs for carbon monoxide). Chest CT showed centrilobular emphysema.
All five had chronic dyspnea,
with progressive functional deterioration in three; and one underÂwent lung
transplantation. Pulmonary histology showed bronchiolitis and alveolar ductitis with B cells, follicles lacking germ centers, and
signifiÂcant emphysema. This pattern was named BADE (Bronchiolitis Alveolar
Ductitis Emphysema). Patients did not
report any previous abnormal occupational exposure.
No cases were identified among
workers from other areas or in the community. Endotoxin conÂcentrations
increased in two samples. Exposure was below occupational limits. Air was
flowing from the machining process of other production areas. The MWF used
developed Pseudomonas pseudoalcaligÂenes and lacked
mycobacterial DNA, but the 16S analysis revealed more bacterial groups. There
was a relationship with the workplace, since all five paÂtients were
specifically involved in production areas. Furthermore, there was an
association with the job, as these previously healthy men experienced an inÂsidious
onset of respiratory symptoms during work. Four were symptomatic outside of the
workplace, and explained they had exacerbation of symptoms while they were at
work. One patient showed a functional improvement during several months when
he/she was outside the area, followed by functional loss upon returning to
their initial tasks.
The researchers’ conclusion would
indicate a previously unrecognized occupational interstitial lung disease.109
To conclude, it is worth
highlighting the work of Petnak and Moua, who conducted a careful analysis of the contributing
factors of HP, commenting on how difficult it is to establish a cause-effect
relationÂship in a problem case. To do so, they formulated a questionnaire
aimed at detecting presumed expoÂsure in individuals presenting with
HP-compatible disease based on four items, namely:
1. Exposure to birds, or items
containing feathers or down.
2. Expression of symptoms at home
or in the workplace.
3. Use of a hot tub, jacuzzi, or sauna.
4. Medical history related to
hobbies or past/curÂrent activities.
The questioning proposed by the
authors would provide support when serology or other clinical and radiological
elements are not irrefutable or if they are unresolved.110
CONCLUSIONS
1. There is an eventuality of suffering
from a work-related or occupational disease in pracÂtically all tasks performed
by the working population.
2. Changes in manufacturing
practices and the addition of novel materials have made occupaÂtional medicine
specialists continue to discover a relationship between new types of exposure
and acute or chronic forms of diffuse parenchymal lung disease.
3. The etiological scope, both
for medical and occupational causes of pulmonary interstitial disease is broad
and permanently enriched with new bibliographic contributions.
4. The complex mechanism of lung
parenchymal repair, manifests itself in the interstitium
with varied responses, both to medical and non-occupational causes, and may
even be different from the same etiology.
5. In order to understand the
link between expoÂsure and disease, occupational medicine specialÂists and
professionals dedicated to safety and hygiene must observe a high index of
suspicion about the potential toxicity of occupational and environmental
manifestations.
REFERENCES
1. Cordier
J. Cryptogenic organizing pneumonia Clin Chest Med.
2004;25:727-38.
https://doi.org/10.1016/j.ccm.2004.06.003
2. Lazor
R. Organizing Pneumonias. En: Cottin V et al. (eds.),
Orphan Lung Diseases: A Clinical Guide to Rare Lung Disease. 2015;363. https://doi.org/10.1007/978-1- 4471-2401-6_24
3.
Okada H, Kurasawa K, Yamazaki R, y col. Clinical features of organizing pneumonia associated with rheumatoid
arthritis. Mod Rheumatol. 2016;26:863-8.
https://doi.org/10.3109/14397595.2016.1153217
4. Ben Saad
A, Joobeur S, Rouetb N. Pneumonie organisĂ©e rĂ©vĂ©Âlatrice d’une polymyosite Pan African Med. J 2014;19:116. https://doi.org/10.11604/pamj.2014.19.116.4939
5.
Ochiai S, Nomoto Y, Yamashita Y, y col. Radiation-induced organizing pneumonia after stereotactic body
radiotheraÂpy for lung tumor. J Radiat Res. 2015;56:904-11. https://doi.org/10.1093/jrr/rrv049
6. Huang P, Kuo
C, Lin C, y col. Mesalazine-related lung disease in a
patient with ulcerative colitis Medicine (Baltimore). 2018;97:e13242. https://doi.org/10.1097/MD.0000000000013242
7.
Barjaktarevic I, Qadir N,
Suri A, y col. Organizing pneuÂmonia as a side
effect of ipilimumab treatment of melaÂnoma. Chest. 2013;143:858-61.
https://doi.org/10.1378/chest.12-1467
8. Watanabe C, Miyata J, Esak K, y col. Pazopanib-induced
organizing pneumonia in a patient with leiomyosarcoma:
A case report. Resp Med Case Rep. 2020;30: 101112. https://doi.org/10.1016/j.rmcr.2020.101112
9. Johar
B, Fatmawati R, Kadir A, y
col. Radiological progresÂsion of COVID-19 organizing pneumonia. Resp Case Rep. 2021;9: e00764
https://doi.org/10.1002/rcr2.764
10. Funk G, Nell C, Pokieser W y col. Organizing pneumoÂnia following Covid19
pneumonia Wien Klin Wochenschr.
2021;133:979-82. https://doi.org/10.1007/s00508-021-01852-9
11.
Zhang Y, Li N, Li Q, y col. Analysis of the
clinical charÂacteristics of 176 patients with pathologically confirmed
cryptogenic organizing pneumonia Ann Transl Med. 2020;763. https://doi.org/10.21037/atm-20-4490
12.
Moriyama H, Kobayashi M, Takada T, y col. Two-dimensionÂal
analysis of elements and mononuclear cells in hard metal lung disease. Am J Respir Crit Care Med. 2007;176:70-7. https://doi.org/10.1164/rccm.200601-134OC
13. Liebow
A, Carrington C. The interstitial pneumonias. In:
Simon M, Potchen EJ, Lemay E, eds. Frontiers in pulmoÂnary
radiology. New York Grune & Stratton,
1969:102-41.
14. Adams T, Butt Y, Batra K, y col. Cobalt related interstiÂtial lung disease. Resp Med. 2017;129:91-7.
https://doi.org/10.1016/j.rmed.2017.06.008
15.
Nunes-Bezerra P, Alves-Vasconcelos A, Albuquerque- Cavalcante L y col. Hard metal lung disease in an oil industry worker. J
Bras Pneumol. 2009;35:1254-8.
https://doi.org/10.1590/S1806-37132009001200015
16.
Carmo-Moreira M, da Rocha Oliveira-Cardoso A, Schuwartz-Tannus D y col. Hard metal pneumocoÂniosis with spontaneous bilateral pneumothorax J
Bras Pneumol. 2010;36:148-15.
https://doi.org/10.1016/j. rmed.2017.06.008
17. Chiarchiaro
J, Tomsica L, Strock S, y
col. A case series describing common radiographic and
pathologic patterns of hard metal pneumoconiosis Respiratory. Med Case
Rep. 2018;25:124-8. https://doi.org/10.1016/j.rmcr.2018.08.006
18. Naqvi
A, Hunt A, Burnett B, y col. Pathologic Spectrum and Lung Dust Burden in Giant
Cell Interstitial PneumoÂnia (Hard Metal Disease/Cobalt Pneumonitis): Review of
100 Cases. Arch Environ & Occup Health. 2008,63:51-70. https://doi.org/10.3200/AEOH.63.2.51-70
19. Hargett
C, Sporn T, Roggli V y col.
giant cell interstiÂtial pneumonia asocciated with nitrofurantoin. Lung. 2006;184:147-9. https://doi.org/10.1007/s00408-005-2574-z
20. Lee B, Balavenkataraman
A, Sanghavi D, y col. Recurrent
nitrofurantoin-induced giant cell interstitial
pneumonia: Case report and literature review. Resp
Med Case Rep. 2015;14:49-52.
https://doi.org/10.1016/j.rmcr.2015.01.002
21. Fernández
PĂ©rez E, Swigris J, Forssen
A, y col. IdentiÂfying an inciting antigen is
associated with improved survival in patients with chronic hypersensitivity
pneuÂmonitis. Chest. 2013;144:1644-51.
https://doi.org/10.1378/chest.12-2685
22. Cormier Y, Schuyler M.
Hypersensitivity pneumonitis and organic dust toxic syndromes. In: Bernstein L,
Chan- Yeung M, Malo J-L,
Bernstein D (eds) Asthma and the workplace, 3rd edn. Marcel Dekker, New York, 2005 p 635-656.
23. Hirschmann
J, Sudhakar N, Pipavath, J,
y col. HypersenÂsitivity Pneumonitis: A Historical, Clinical, and RadioÂlogic
Review. RadioGraphics. 2009;29:1921-38.
https://doi.org/10.1148/rg.297095707
24. Raghu G, Remy-Jardin M, Ryerson C, y col. Diagnosis of Hypersensitivity
Pneumonitis in Adults An Official ATS/JRS/ALAT Clinical Practice Guideline. Am
J Resp Crit Care Med. 2020;202:e36-e69. https://doi.org/10.1164/rccm.202005-2032ST
25.
Yoshida K, Suga M, Nishiura Y, y col. Occupational hyperÂsensitivity pneumonitis in Japan: data on a
nationwide epidemiological study. Occup Environ Med.
1995;52:570-4. https://doi.org/10.1136/oem.52.9.570
26. Moon Bang K, Weissman D, Pinheiro G, y col.
Twenty- Three Years of Hypersensitivity Pneumonitis MortalÂity Surveillance in
the United States American. Am J Ind Med. 2006;49:997-1004. https://doi.org/10.1002/ajim.20405
27. Salvaggio
J. Hypersensitivity Pneumonitis: Pandora ’s Box. New Engl J Med. 1970;283:514-5.
https://doi.org/10.1056/NEJM197008062830610
28. Campbell J. Acute Symptoms
Following Work with Hay. Brit Med J. 1932;2:1143-4.
29. Fawcitt R. Occupational diseases of lungs in agriculÂtural
workers. Brit J Radiol. 1938;11:378-92.
https://doi.org/10.1259/0007-1285-11-126-378
30. Liu S, Chen D, Fu S, y col.
Prevalence and risk factors for farmer’s lung in greenhouse farmers: an
epidemiological study of 5,880 farmers from Northeast China. Cell
BioÂchem Biophys.
2015;71:1051-7.
https://doi.org/10.1007/s12013-014-0308-7
31. Gbaguidi-Haore
H, Roussel S, Reboux G, y
col. MultiÂlevel analysis of the impact of environmental factors and
agricultural practices on the concentration in hay of microorganisms
responsible for farmer’s lung disease. Ann Agric
Environ Med. 2009;16:219-225.
32. Dickie
H, Rankin J. Farmer’s lung an acute granulomaÂtous interstitial pneumonitis
occurring in agricultural workers JAMA. 1958;167:1069-78.
https://doi.org/10.1001/jama.1958.02990260011004
33. Peppys
J, Jenkins P. Precipitin (F.L.H.) Test in Farmer’s Lung Thorax. 1965;20:21-36. https://doi.org/10.1136/thx.20.1.21
34.
Hapke E, Seal M, Thomas G y
col. Farmer’s lung A cliniÂcal, radiographic, functional,
and serological correlation of acute and chronic stages Thorax. 1968;23:451-69. https://doi.org/10.1136/thx.23.5.451
35. Cuthbert O, Gordon M. Ten
year follow up of farmers with farmer’s lung. Br J Ind
Med. 1983;40:173-6.
https://doi.org/10.1136/oem.40.2.173
36. Depierre
A, Dalphin J, Pernet D y
col. Epidemiological study of farmer’s lung in five districts of the French
Doubs province. Thorax. 1988;43:429-35.
https://doi.org/10.1136/thx.43.6.429
37. Soumagne
T, Pana-Katatali H, Degano
B, y col. Combined pulmonary fibrosis and emphysema in hypersensitivity
pneumonitis BMJ Case Rep. 2015. https://doi.org/10.1136/bcr-2015-211560
38. Di Martino P.
Ways to improve biocides for metalworkÂing fluid. AIMS Microbiol. 2021;7:13-27.
https://doi.org/10.3934/microbiol.2021002
39. Gupta A, Rosenman
K. Hypersensitivity Pneumonitis Due to Metal Working Fluids: Sporadic or Under
Reported? Am J Ind Med. 2006;49:423-33.
https://doi.org/10.1002/ajim.20312
40. Bakerly
N, Moore V, Vellore A, y col. Fifteen-year trends in occupational asthma: data
from the shield surveillance scheme. Occup Med (Lond). 2008;58:169-74. https://doi.org/10.1093/occmed/kqn007
41. Rosenman
K, Reilly M, Kalinowski D. Annual report on
work-related asthma in Michigan. East Lansing, Michigan (11/13/07); 2006
http://www.oem.msu.edu/asthma/06WRA_all.pdf (accessed 10/29/2008)
42. Hendy M, Beattie B, Burge P.
Occupational asthma due to an emulsified oil mist. Br J Ind
Med. 1985;42:51-4. https://doi.org/10.1136/oem.42.1.5164
43. Robertson A, Weir D, Burge P.
Occupational asthma due to oil mists. Thorax. 1988;43:200-5. https://doi.org/10.1136/ thx.43.3.200
44. Massin
N, Bohadana A, Wild P, y col. Airway responÂsiveness,
respiratory symptoms, and exposures to soluble oil mist in mechanical workers. Occup EnÂviron Med. 1996;53:748-52.
https://doi.org/10.1136/oem.53.11.748
45. Greaves I, Eisen E, Smith T, y col. Respiratory Health of Automobile
Workers Exposed to Metal-Working FluÂid Aerosols: Respiratory Symptoms Am J Ind Med. 1997;32:450-9.
https://doi.org/10.1002/(SICI)1097-0274(199711)32:5<450::AID-AJIM4>3.0.CO;2-W
46. Bernstein D, Lummus Z, Santilli G, y col. Machine opÂerator’s lung a hypersensitivity
pneumonitis disorder associated with exposure to metalworking fluids aeroÂsols.
Chest. 1995; 1008:636-41.
https://doi.org/10.1378/chest.108.3.636
47. Hodgson M, Bracker A, Yang C, y col. Hypersensitivity pneumonitis in a
metalworking environment. Am J Ind Med. 2001;39:616-8. https://doi.org/10.1002/ajim.1061
48. Fox J, Anderson H, Moen T, y
col. Metalworking fluid-associated hypersensitivity pneumonitis: an outbreak
investigation and case–control study. Am J Ind Med.
1999;35:58-67. https://doi.org/10.1002/(SICI)1097-0274(199901)35:1<58::AID-AJIM8>3.0.CO;2-5
49. Bracker
A, Storey E, Yang C et al. An outbreak of hyÂpersensitivity
pneumonitis at a metalworking plant: a longitudinal assessment of intervention
effectiveness. Appl Occup
Environ Hyg. 2003;18:96-108.
https://doi.org/10.1080/10473220301436
50. Gordon T, Nadziejko
C, Galdanes K, y col. Mycobacterium immunogenum causes hypersensitivity pneumonitis-like
pathology in mice. Inhal Toxicol. 2006;18:449-56.
https://doi.org/10.1080/08958370600563904
51. Weiss L, Lewis R, Rossmoore H y col. Respiratory illness in workers exposed
to Metalworking fluid contaminated with nontuberculous
mycobacteria: Ohio, 2001. MMWR 2002;51:349-52.
52. Shelton B, Flanders D, Morris
G. Mycobacterium sp. as a possible cause of hypersensitivity pneumonitis in maÂchine
workers. Emerg Infect Dis. 1999;5:270-3.
https://doi.org/10.3201/eid0502.990213
53. Cullinan
P, Lowson D, Nieuwenhuijsen
M, y col. Work related symptoms, sensitisation, and
estimated expoÂsure in workers not previously exposed to flour. Occup Environ Med. 1994;51:579-83.
https://doi.org/10.1136/oem.51.9.579
54. Brant A. Baker’s asthma. Curr Opin Allergy Clin Immunol. 2007;7:152-5.
https://doi.org/10.1097/ACI.0b013e328042ba77
55. Simonis
B, Hölzel C, Stark U. Glucoamylase:
a current allergen in the baking industry Allergo J
Int. 2014;23:269- 73.
https://doi.org/10.1007/s40629-014-0034-0
56. Diederichs
W, Lubbers P. [Flour asthma as an occupational disease] (German). Zentralbl Arbeitsmed 1955;5:189-97.
57. Thiel H, Ulmer W. Bakers’
Asthma: Development and Possibility for Treatment Chest. 1980;78:400-6.
https://doi.org/10.1378/chest.78.2_Supplement.400
58. Gerfaud-Valentin
M, Reboux G, Traclet J, y
col. OccuÂpational Hypersensitivity Pneumonitis in a Baker. Chest.
2014;14:856-8. https://doi.org/10.1378/chest.13-1734
59. Lunn
J, Hughes T. Pulmonary Hypersensitivity to the Grain Weevil. Br J Ind Med. 1967;24:158-61.
https://doi.org/10.1136/oem.24.2.158
60. van Heemst
R, Sander I, Rooyackers J, y col. HypersenÂsitivity
pneumonitis caused by occupational exposure to phytase.
Eur Respir J. 2009;33:1507-9. https://doi.org/10.1183/09031936.00035408
61. Brant A, Berriman
J, Sharp C, y col. The changing distriÂbution of occupational asthma: a survey
of supermarket bakery workers. Eur Respir J. 2005;25:303-8.
https://doi.org/10.1183/09031936.05.00054004
62. Sullivan A, Shrestha P, Lanham T, y col. Bird Fancier’s lung: An
underdiagnosed etiology of dyspnea. Respir Med Case
Rep. 2020;31: 101288
https://doi.org/10.1016/j.rmcr.2020.101288
63. Christensen L, Duwayne Schmidt C, Robbins L. PiÂgeon breeders’ disease: a
prevalence study and reÂview. Clin Allergy. 1975;5:417-30.
https://doi.org/10.1111/j.1365-2222.1975.tb01881.x
64. Morell
F, Roger A, Reyes L, y col. Bird Fancier’s Lung A Series of 86 Patients
Medicine. 2008;87:110-30. https://doi.org/10.1097/MD.0b013e31816d1dda
65.
Rodrigo M, Benavent M, Cruz M, y col. Detection of speÂcific antibodies to pigeon serum and bloom antigens by
enzyme linked immunosorbent assay in pigeon breeder’s
disease. Occup Environ Med. 2000;57:159-64.
https://doi.org/10.1136/oem.57.3.159
66. Shiroshita
A, Tanaka Y, Nakashima K, y col. Diagnostic accuracy of specific IgG antibodies for bird fancier’s lung: a systematic review
and meta-analysis Ann Transl Med. 2019;7:655.
https://doi.org/10.21037/atm.2019.10.65
67. McSharry
C, Dye G, Ismail T, y col. Quantifying serum antibody in bird fanciers’
hypersensitivity pneumonitis. BMC Pulm Med. 2006;6:16 https://doi.org/10.1186/1471-2466-6-16
68. Chopra V, Lal
Joshi J, Mrigpuri P, y col. Pigeon fancier’s lung –
An under-diagnosed cause of severely debilitatÂing and chronic breathlessness
Egyptian J Dis Tuberc. 2017;66:557-9.
https://doi.org/10.1016/j.ejcdt.2016.08.002
69. Cooper C, Teleb
M, Elhanaf S, y col. Bird fanciers’ lung induced by
exposure to duck and goose feathers. Am J Case Rep. 2014;15:155-8.
https://doi.org/10.12659/AJCR.890184
70. Blake B, Mc
Kay J, Rainey H, y col. Pulmonary Opacities Resulting from Di-isocyanate Exposure. J Coll Radiol Aust. 1965;9:45-8.
https://doi.org/10.1111/j.1440-1673.1965.tb00974.x
71. Bernstein D, Korbee L, Staude T, y col. The
low prevaÂlence of occupational asthma and antibody-dependent sensitization to diphenylmethane diisocyanate in a
plant engineered for minimal exposure to disocyanates.
J Allergy Clin Immunol.
1993;92:387-96. https://doi.org/10.1016/0091-6749(93)90117-X
72. Musk A, Peters J, Wegman D. lsocyanates and
Respiratory Disease: Current Status. Am J Ind Med
1988;13:331-49.
https://doi.org/10.1002/ajim.4700130304
73. Baur
X. Hypersensitivity pneumonitis (extrinsic allergic alveolitis)
induced by isocyanates. J Allergy
Clin ImÂmunol. 1995;95:1005-10. https://doi.org/10.1016/S0091-6749(95)70101-X
74.
Vandenplas O, Malo JL, Dugas
M, y col. HypersensiÂtivity pneumonitis-like reaction among
workers exÂposed to diphenylmethane diisocyanate (MDI). Am Rev Respir
Dis. 1993;147:338-46.
https://doi.org/10.1164/ajrccm/147.2.338
75. Schreiber J, Knolle J, Sennekamp J, y col.
Sub-acute ocÂcupational hypersensitivity pneumonitis due to low-level exposure
to diisocyanates in a secretary. Eur Respir
J. 2008;32:807-11.
https://doi.org/10.1183/09031936.00060507
76.
Suzuki N, Matsuzaki G, Arai
Y, y col. A case of hyperÂsensitivity pneumonitis in which serum
specific antiÂbodies for three species of isocyanate
molecules were demonstrated. Nihon Kyobu Shikkan Gakkai Zasshi. 1992;30:478-84.
77. Tabata
H, Mochizuki Y, Nakahara Y, y col. lHypersensiÂtivity
pneumonitis caused by isocyanate exposure during
recreational painting. Nihon Kokyuki Gakkai Zasshi. 2009;47:1002-7.
78. Musk W, Peters J, Wegman D. Isocyanates and
Respiratory Disease: Current Status. Am J Ind Med
1988;13:331-49.
https://doi.org/10.1002/ajim.4700130304
79. Nakashima K, Takeshita T, Morimoto K. Diagnosis and Prevention of
Diseases Induced by Isocyanate. Environ Health Prev Med 2002;7:40-6.
https://doi.org/10.1007/ BF02897329
80. Smilkstein
M, J, Burton B; T, Keene W y col. Acute respiratory illness linked to use of
aerosol leather condiÂtioner, Oregon, 1992. JAMA. 1993;269:568-9.
https://doi.org/10.1001/jama.1993.03500050028008
81.
Testud F, Gabrielle L, Paquin
M y col. Alvéolite aiguë après utilisation d’un aérosol imperméabilisant : À propos de deux observations. Rev Méd Inteme. 1998;19:262-4. https://doi.org/10.1016/S0248-8663(97)89328-9
82. Burkhart K, Britt A, Petrini G, y col. Pulmonary ToxÂicity Following Exposure to
an Aerosolized Leather Protector. Clin Toxicol.1996;34:21-4. https://doi.org/10.3109/15563659609020228
83. Wallace G, Brown P. Horse rug
lung: toxic pneumonitis due to fluorocarbon inhalation. Occup
Environ Med. 2005;62:414-16.
https://doi.org/10.1136/oem.2004.015784
84. Scheepers
P, Masen-Poos L, van Rooy
F, y col. Pulmonary injury associated with spray of a
water-based nano-sized waterproofing product: a case
study. J Occup Med Toxicol
2017;12:33. https://doi.org/10.1186/s12995-017-0180-7
85. Tan D, Wang C, Ling J, y col.
Acute respiratory disÂtress syndrome after accidental inhalation of fluoÂrocarbon
monomers and pyrolysis products. Occup Environ Med.
2016;73:287-8.
https://doi.org/10.1136/oemed-2015-103185
86. Vernez
D, Bruzzi R, Kupferschmidt
H, y col. Acute ReÂspiratory Syndrome After Inhalation
of Waterproofing Sprays: A Posteriori Exposure-Response Assessment in 102
Cases. J Occup Environ Hyg.
2006;3:250-61.
https://doi.org/10.1080/15459620600628845
87. Walters G, Trotter S, Sinha B, y col. Biopsy-proven hypersensitivity pneumonitis
caused by a fluorocarbon waterproofing spray Occupational Medicine 2017;
67:308- 310. https://doi.org/10.1093/occmed/kqx039
88. Shirai
T, Sato A, Hara Y. Epigallocatechin gallate. The major causative agent of
green tea-induced asthma. Chest. 1994;106:1801-5. https://doi.org/10.1378/chest.106.6.1801
89. Uragoda
C. Tea maker’s asthma. Brit J Industr Med. 1970;27:181-2. https://doi.org/10.1136/oem.27.2.181
90. Cartier A, Malo JL. Occupational asthma due to tea
dust. Thorax 1990;45:203-6.
https://doi.org/10.1136/thx.45.3.203
91.
Sano T, Gemma H, Satou M y
col. A case of hypersensitivÂity pneumonitis in a tea bag
manufacturer employee (in Japanese). Nihon Kyobu Rinsho 2004;63:393-398.
92. Otera
H, Tada K, Sakurai T, y col. Hypersensitivity pneumonitis associated with
inhalation of catechin-rich green tea extracts.
Respiration 2011;82:388-92.
https://doi.org/10.1159/000324450
93. Tanaka Y, Shirai
T, Enomoto N y col. Occupational hypersenÂsitivity
pneumonitis in a green tea manufacturer. Respirol
Case Rep. 2016;4:e00152.
https://doi.org/10.1002/rcr2.152
94. Shirai
T, Sato A, Hara Y. Epigallocatechin gallate. The major causative agent of
green tea-induced asthÂma. Chest. 1994;106:1801-5. https://doi.org/10.1378/chest.106.6.1801
95. Shirai
T, Sato A, Chida K, y col. Epigallocatechin
gallate-induced histamine release in patients with
green tea-inÂduced asthma. Ann Allergy Asthma Immunol. 1997;79:65- 9.
https://doi.org/10.1016/S1081-1206(10)63087-6
96. Avila R, Villar
T. Suberosis: respiratory disease in cork workers. Lancet. 1968;1:620-1. https://doi.org/10.1016/S0140-6736(68)91239-7
97.
Ruiz Manzano J, Valdés M, León M, y col. Suberosis:
alveolitis alĂ©rgica extrĂnseca en un trabajador del corcho. Med Clin (Barc). 1984;82:265-26.
98. Campbell J, Kryda M, Treuhaft M, y col. Cheese
Worker’s Hypersensitivity Pneumonitis. Am Rev Respir
Dis. 1983;127:495-6.
https://doi.org/10.1164/arrd.1983.127.4.495
99. Galland
C, Reynaud R, De Haller B, y col. Cheese-washer’s disease. A
current stable form of extrinsic allergic alveoÂlitis
in a rural setting. Rev Mal Respir. 1991;8:381-6.
100. Bringgold
W, Halliday S. The cheese whiz lung: hypersenÂsitivity
pneumonitis in an imitation cheese factory worker.
https://doi.org/10.1016/j.chest.2020.08.1578
101. Banaszak
E, Thiede W, Fink J. Hypersensitivity pneuÂmonitis
due to contamination of an air conditioner. New Engl
J Med 1970;283:271-6.
https://doi.org/10.1056/NEJM197008062830601
102. Hodgson M, Morey P, Simon J,
y col. an outbreak of recurÂrent acute and chronic hypersensitivity pneumonitis
in office workers. Amer J Epidemiol
1987;125:631-8.
https://doi.org/10.1093/oxfordjournals.aje.a114576
103. Nakazawa
T, Tochigi T. Hypersensitivity pneumonitis due to Mushroom (Pholiota
nameko) spores. Chest. 1989;95:1149-51. https://doi.org/10.1378/chest.95.5.1149
104. Saikai
T, Tanaka H, Fuji M, y col. Hypersensitivity pneuÂmonitis due by the spore of Pleurotus eryngii. Intern Med
2002;41:571-3.
105. Moore J, Convery
R, Milla C, y col. Hypersensitivity Pneumonitis
Associated with Mushroom Worker’s Lung: An Update on the Clinical Significance
of the Importation of Exotic Mushroom Varieties. Int
Arch Allergy Immunol 2005;136:98-102.
https://doi.org/10.1159/000083298
106. Yoshikawa S, Tsushima K,
Koizumi T, y col. HypersensitivÂity Pneumonitis Induced by Spores of Penicillium citrinum in a Worker
Cultivating Enoki Mushroom.
https://doi.org/10.2169/internalmedicine.45.1646
107.
Ampere A, Delhaes L, Soots
J, y col. Hypersensitivity pneumonitis induced by Shiitake
mushroom spores. Med Mycol. 2012;50:654-7.
https://doi.org/10.3109/13693786.2012.658091
108. Marchisio
V, Sulotto G, Botta A, y
col. Aerobiological analysis in a salami factory: a possible case of extrinÂsic
allergic alveolitis by Penicillium
camembertii. Med Mycol 1999;37:285-9. https://doi.org/10.1080/j.1365- 280X.1999.00231.x
109. Cummings K, Stanton M, Nett R, y col. Severe lung disÂease
characterized by lymphocytic bronchiolitis, alveolar ductitis,
and emphysema (BADE) in industrial machine manufacturing workers. Am J Ind Med. 2019;62:927-37.
https://doi.org/10.1002/ajim.23038
110. Petnak
T, Moua T. Exposure assessment in hypersensitivÂity
pneumonitis: a comprehensive review and proposed screening questionnaire ERJ
Open Res 2020;6:00230- 2020.
https://doi.org/10.1183/23120541.00230-2020