Autor :Ubal, Leonardo German1-2, Acosta, MarĂa Alejandra1, Oviedo, Eduardo Enrique1, Fernández, RocĂo Guadalupe1, Appiolaza, Alejandra1, Kevorkof, Gregorio Varujan1-2
1 Hospital Tránsito Cáceres de Allende (HTCA), Pulmonology Service, Córdoba Capital City, Córdoba, Argentina.
2 Chair of Clinical Medicine II, U.H.M.I. N° 5 of HTCA, Faculty of medical Sciences - Universidad Nacional de Córdoba (UNC), Argentina.
https://doi.org/10.56538/ramr.HYAU6023
Correspondencia : Leonardo Ubal e-mail: leoub6@hotmail.com
ABSTRACT
Introduction: SARS-CoV-2 is likely to favor the transition from infection to tuberculous disease. Although information is limited, there
is progress in understanding the interÂaction between COVID-19 and
tuberculosis. New investigations yielded unexpected similarities in the
pathogenesis and evolution of the coinfection.
Prolonged lymphopenia, hyperinflammation,
lung tissue injury, and imbalance in CD4+ T-cell subsets associated with
COVID-19 could propagate M. tuberculosis infection and disease
progression.
Case reports: we present three young patients, without comorbidities, with risk facÂtors
for latent tuberculous infection, diagnosed with
pulmonary tuberculosis post mild COVID-19, with symptomatic treatment (not
corticosteroids).
Discussion: these cases raise the probable impact of SARS-CoV-2 in the transition
from latent tuberculous infection to disease,
excluding the already proven influence of corticosteroids and severe forms of
COVID-19. There is increasing evidence to support this idea.
Key words: COVID-19, SARS-CoV-2, Tuberculosis
RESUMEN
Introducción: es probable que SARS-CoV-2 favorezca el paso de infección a enfermeÂdad
tuberculosa. Si bien la información es limitada, hay avances en la
comprensión de la interacción COVID-19 y Tuberculosis. Nuevas
investigaciones arrojaron similitudes inesperadas en la patogenia y
evolución de la coinfección. Linfopenia prolongada, hipeÂrinflamación,
lesión del tejido pulmonar y desequilibrio en los subconjuntos de
células T CD4+ asociados con COVID-19, podrían propagar la
infección por M. tuberculosis y progresión de la
enfermedad.
Casos clínicos: presentamos tres pacientes jóvenes, sin comorbilidades, con factores
de riesgo para Infección Tuberculosa Latente, diagnosticados de Tuberculosis
pulmonar posterior cursado COVID-19 leve, de tratamiento sintomático (no
corticoideo).
Discusión: estos casos plantean el probable impacto del SARS-CoV-2 en el paso de
Infección Tuberculosa Latente a enfermedad, excluyendo la ya demostrada
influencia de los corticoides y formas graves de COVID-19. Existe cada vez
más evidencia que refuerza esta idea.
Palabras clave: COVID-19, SARS-CoV-2, Tuberculosis
Received: 07/29/2022
Accepted: 11/30/2022
INTRODUCTION
In December 2019, the world faced
a new coronaÂvirus (SARS-CoV-2) which caused COVID-19.1
In 2020, as a result of the pandemic, the tuberculosis (TB)
control services were interrupted. The World Health Organization (WHO) recorded
a reduction in the global number of patients diagnosed and treated for TB and
an increase in the number of deaths, for the first time in decades.2, 3
COVID-19 can affect people
infected or ill with TB before, during or after being cured, facilitating in
some cases the transition from latent tubercuÂlous
infection (LTBI) to disease, and also increasÂing the possibility of making TB
evolution more severe due to a higher extension of pulmonary lesions.3-5
It has been proven that people
with TB have higher risk of death from COVID-193 and that the use of
corticosteroids both for the acute phase and for post COVID-19 organizing
pneumonia can lead to TB reactivation.1, 2, 5 It has also been observed that the
“unfavorable” evolution of COVID-19 implies a higher risk of progression from
LTBI to active TB.6
Even though information is still
limited, there is growing understanding of the interaction of both diseases,
and COVID-19 will probably favor the transition from infection to tuberculous disease, regardless of the severity of its
course.7
New research on the molecular and
cellular mechanisms of M. tuberculosis and SARS-CoV-2 infections have
yielded unexpected similarities regarding the pathogenesis and evolution of the
coinfection. Long-term lymphopenia,
hyperinÂflammation, lung tissue injury, and imbalance
in CD4+ T-cell subsets associated with COVID-19 could propagate the M.
tuberculosis infection and disease progression.1, 8
The co-existence of TB and
COVID-19 is preÂsented in Argentina with undetermined values, and a risk which
we can assume has increased.2,
9, 10
We present three case reports of
young patients without relevant comorbidities, with risk factors for LTBI
(healthcare personnel, family contact) who were diagnosed with pulmonary TB
post mild COVID-19 infection, with outpatient symptomatic treatment (not
corticosteroids).
CASE REPORTS
Case N° 1: 25-year-old female, resident doctor. Diagnosis of prolactinoma,
medicated with cabÂergoline. Claims she doesn’t have
any history of allergies, surgeries, or use of toxic substances. No family
members or cohabitants with history of TB. Complete vaccination scheme for
SARS-CoV-2; BCG (bacille Calmette-Guerin)
administered when the patient was a child. She had COVID-19 in May 2021, with
mild symptoms of the upper airway (UAW), cephalea,
dry cough, anosmia and ageusia; no pulmonary
involvement.
After having COVID-19, she still
had dry cough, initially associated with post-viral sequelae.
In July 2021, she started to have episodes of night sweating, weight loss and
occasional sibilance, so she started to use supplementary methods, and the
chest X-ray showed cavitary infiltrate in right upper
lobe (RUL), with suspicion of pulmonary TB. Respiratory physical examination: crepitant rales in the upper area
of the right field and isolated sibilance at auscultation.
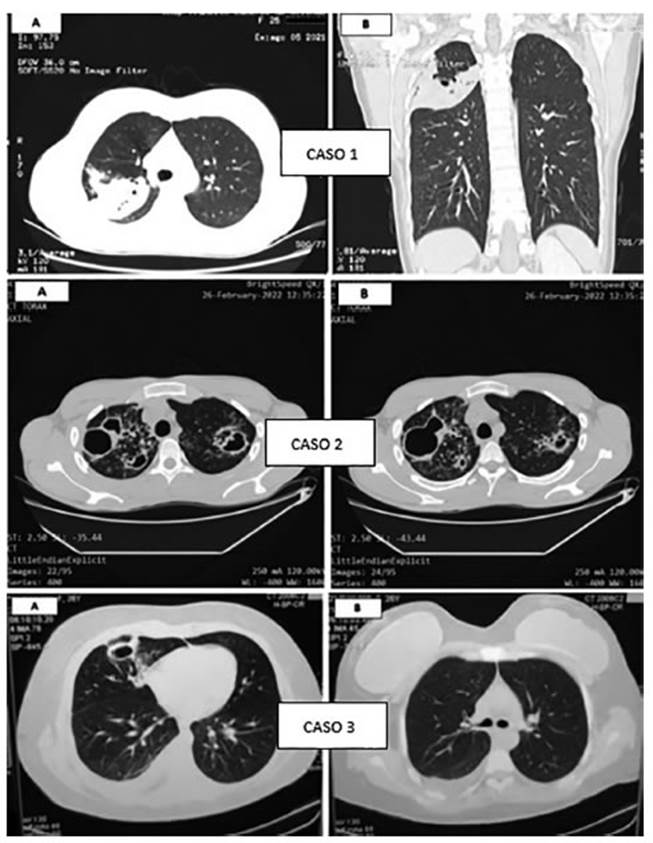
August 2021: chest CAT (computed
axial tomogÂraphy) shows condensation with alveolar aspect at the posterior
segment of the RUL, identifying small images compatible with caves, the biggest
beÂing 8 mm (Figure 1). Laboratory: high erythrocyte sedimentation rate,
negative viral serology. DiagÂnostic fibrobronchoscopy
(FBC) performed with lavage and bronchial biopsy with 3 and 8 BAAR
(acid-alcohol-resistant bacillus)/field, respectively, and positive cultures.
Patient begins treatment for Category 1 pulmonary TB (unilateral with cave),
first phase 2HRZE (isoniazid, H; rifampicin, R; pyrazinamide, Z, and ethambutol, E), and second phase 4HR (isoniazid, H;
rifampicin, R), with faÂvorable response.
Case N° 2: 22-year-old male who works at an energy cooperative. No history of
diseases, allergies or use of toxic substances. Cohabits with mother, father,
and 3 siblings. History of pulmonary TB in the family: the patient’s mother in
2014 (full treatÂment), and also the patient’s paternal grandfather and uncle.
Complete vaccination scheme for SARS-CoV-2; BCG administered when the patient
was a child. He had COVID-19 in April 2021, without respiratory symptoms.
Since May 2021, after COVID-19,
he’s been having cough and mucous expectoration, occasionÂally mucopurulent (not hemoptoic),
dyspnea on exertion, and weight loss. Makes several medical consultations and
uses different antibiotic regiÂmens without symptom improvement. Respiratory
physical examination: rhonchus and movement of secretions with coughing.
February/March 2022: chest CAT
shows mulÂtiple caves of thick walls predominant in upper fields, associated
with large tree-in-bud pattern and ill-defined opacities with consolidative
aspect (Figure 1). Laboratory: high erythrocyte sedimenÂtation rate, negative
viral serology. Sputum for Koch with >10 BAAR/field in two samples. PaÂtient
begins treatment for Category 1 pulmonary TB (bilateral with cave), first phase
2HRZE and second phase 4HR.
Case N° 3: 26-year-old female, resident doctor. History of obesity class II (BMI:
35). No history of allergies, surgeries, or use of toxic substances. No family
members or cohabitants with history of TB. Complete vaccination scheme for
SARS-CoV-2; BCG administered when the patient was a child. She had COVID-19 in
January 2022 with mild symptoms; no pulmonary involvement.
After having COVID-19, she still
had asthenia, and in March she also had fever and cough with mucous
expectoration. Respiratory physical examiÂnation: isolated rhonchus at
auscultation.
March 2022: chest CAT shows subsegmental linear atelectasis that affect the medial
segment of the right middle lobe, apart from thick caves and mild homolateral pleural effusion (Figure 1). Laboratory: high
erythrocyte sedimentation rate, negative viral serology. Negative sputum bacillosÂcopies, positive cultures/GeneXpert® low, without
resistance to rifampicin. Patient begins treatment for Category 1 pulmonary TB
(unilateral with cave + pleurisy), first phase 2HRZE and second phase 4HR.
DISCUSSION
The presentations of these case
reports raise the probable impact of SARS-CoV-2 in the transition from LTBI to
disease, excluding the already proven influence of corticosteroids and severe
forms of COVID-19. There is increasing evidence to supÂport this idea.
TB affects mainly the lungs when
the adaptive immune response, mostly performed by T cells, is altered. With
coronavirus infection, there is inÂcreased depletion of T cells and a decline
in their functional diversity. According to several studies, viruses have been
found in T lymphocytes, macÂrophages and dendritic cells that can also alter
their function. So, the coronavirus infection, which activates cellular
immunity, results in the depleÂtion of the system that is used for fighting TB1.
The significant influence of the
SARS-CoV-2 virus on the immune system that produces severe immunosuppression,
activation and progression of existing TB foci can modify the tuberculous infecÂtion due to changes in the nature and
intensity of the local cellular immune response. Just like it happens with HIV
infections at the AIDS stage, when the reactions of lymphocytes, of epithelioid cells and giant cells become less intense, and
inÂflammation mechanisms and quick dissemination of TB predominate1.
The TB/COVID-19 Global Study
Group7 found 71 patients (out of 767) who had been diagnosed with
COVID-19 before TB; 48% showed caves, a condition that will likely develop in
more than 30 days (time sufficient to develop the disease). Thus, this indirect
evidence is against the preÂsumption.
A South African study8
showed that COVID-19 didn’t trigger the concomitant activation of CD4+ T cells
specific of M. tuberculosis; this is against the hypothesis. However, a
significant reduction was found in the frequency of these cells in COVID-19
patients compared to pre-pandemic healthy parÂticipants with LTBI. This
reduction could affect the host’s capacity to control the infection with M.
tuberculosis (latent or new).
There is still a very long way to
go, and these questions have been raised. Additional longituÂdinal studies that
observe patients with TB and COVID-19 over time and compare the proportion of
those who acquire the TB disease with a control group without COVID-19 can
provide a better understanding of their interaction.7, 11
In general, data suggest that TB
and COVID-19 are a “cursed duet” and require immediate care7.
Conflict of interest
Authors declare there isn’t any
conflict of interest in relaÂtion to the contents of this article.
REFERENCES
1. Starshinova
A, Kudryavtsev I, Malkova
A, et al. Molecular and Cellular Mechanisms of M. tuberculosis and SARS-CoV-2
Infections - Unexpected Similarities of Pathogenesis and What to Expect from
Co-Infection. Int J Mol
Sci. 2022;23:2235. https://doi.org/10.3390/ijms23042235
2. Brian MC. Tuberculosis y COVID-19. Asociación Argentina de Medicina
Respiratoria - AAMR. La Gaceta. Julio 2020. Disponible en
http://www.aamr.org.ar/lagaceta/tuberculoÂsis-y-covid-19/
3. Ruhwal
M, Hannay E, Sarin S, Kao
K, Sen R, Chadha S.
Considerations for simultaneous testing of COVID-19 and tuberculosis in
high-burden countries. Lancet Glob Health.
2022;10:e465-e466.
https://doi.org/10.1016/S2214-109X(22)00002-X
4. Comella del Barrio P, De
Souza Galvao ML, Prat AymÂerich
C, Dominguez J. Impacto de la COVID-19 en el conÂtrol
de la tuberculosis. Arch Bronconeumol.
2021;57:5–6.
https://doi.org/10.1016/j.arbres.2020.11.016
5. Viscaa D, Ongc C, Tiberie S, et al.
Interacción Tuberculosis y COVID-19: una revisión de efectos
biológicos, clínicos y salud pública. Pulmonology.
2021; 27: 151-65. https://doi.org/10.1016/j.pulmoe.2020.12.012
6. Rodrigo T, Gullón JA, Tabernero E, et al.
Impacto de la pandemia COVID-19 en el Programa Integrado de
Investigación en Tuberculosis (PII-TB) de la Sociedad Española de
Neumología y Cirugía Torácica (SEPAR). EnfEmerg. 2022;21:81-4. Available at
http://www.enfermedadesemergentes.com/articulos/a818/4_origiÂnal_breve_rodrigo.pdf
7. The TB/COVID-19 Global Study
Group. Tuberculosis and COVID-19 co-infection: description of the global cohort.
Eur Respir J. 2022; 59:
2102538. https://doi.org/10.1183/13993003.02538-2021.
8. Riou
C, Bruyn E, Stek C, et al.
Relationship of SARS-CoV- 2-specific CD4 response to
COVID-19 severity and impact of HIV-1 and tuberculosis coinfection.
J Clin Invest.
2021; 131:e149125. https://doi.org/10.1172/JCI149125
9. Vanzetti C, Salvo C, Kuschner
P, Brusca S, Solveyra F, Vilela
A. Coinfección Tuberculosis y COVID-19. MEÂDICINA
(Buenos Aires). 2020;80:100-3. Available
at https://www.medicinabuenosaires.com/indices-de-2020/
volumen-80-ano-2020-s-6-indice/coinfeccion_tuberculoÂsis/
10. Palmero D, Levi A, Casco N, et al. COVID-19 y
tuberculoÂsis en 5 hospitales de la Ciudad de Buenos Aires. Rev Am Med Resp. 2020;3:251-4. Avaiable at
https://www.ramr.org/articulos/volumen_20_numero_3/comunicacion_breve/comunicacion_breve_covid-19_y_tuberculosis_en_5_hospiÂtales_de_la_ciudad_de_buenos_aires.pdf
11. Saunders M, Evans C. COVID-19, tuberculosis y
pobreza: preÂvenir una tormenta perfecta. Eur Resp J. 2020;56:2001348. https://doi.org/10.1183/13993003.01348-2020