Autor : Borsini, Eduardo1, Blanco, MagalÃ1, Ernst, Glenda2, Robaina, Gabriela2, Sills, Nora3, Bosio, MartÃn2, Salvado, Alejandro2, Rabec, Claudio4
1Sleep and Ventilation Unit, Hospital BritÃĄnico de Buenos Aires, Argentina. 2Department of Respiratory Medicine, Hospital BritÃĄnico de Buenos Aires, Argentina. 3Kinesiology Service, Hospital BritÃĄnico de Buenos Aires, Argentina. 4Service de Pneumologie et RÃĐanimation Respiratoire, Centre Hospitalier et Universitaire de Dijon, France.
https://doi.org/10.56538/ramr.DAVA2524
Correspondencia : Dr. Eduardo Borsini, HosÂpital BritÃĄnico, Perdriel 74, Buenos Aires, Argentina; (CP1280AEB) Telephone number: +5411-43096400 Ext. number: 2808. Cell phone: +5491153341951 E-mail: borsinieduardo@yaÂhoo.com.ar - eborsini@hbritanico.com.ar Funding: this work didnât receive financial support.
ABSTRACT
Introduction: Clinical experience has allowed the use of non-invasive ventilation outÂside
the acute-care setting. We describe the clinical profile and evolution of
patients who received non-invasive ventilation in a regular ward.
Materials and methods: Retrospective study in patients with ventilatory
support for one year in a general hospital.
Results: Non-invasive ventilation was delivered to 43 patients, 67.4% of which
had hyÂpercapnia. The male/female ratio was 1:1. Age and
BMI (Body Mass Index) were 68.3 Âą 12.4 years and 30.1 Âą 12.3 kg/m2, and
the main diagnoses were chronic obstructive pulmonary disease, neuromuscular
disease and obesity-hypoventilation. One third of patients began non-invasive
ventilation in the Intensive Care Unit, and two thirds had been using
non-invasive ventilation at their homes before being admitted with exacerbaÂtion
of chronic obstructive pulmonary disease (39.5%) or disease progression (14%).
Hospital length of stay was 12.1 Âą 7 d (14 Âą 9 in survivors and 5.7 Âą 3 in
deceased paÂtients). Arterial blood gas analysis on admission showed: PaCO2 (partial
pressure of arÂterial carbon dioxide), 52.7 Âą 13.7 mmHg; PaO2 (partial pressure of arterial oxygen), 72.2 Âą
16.2 mmHg, and pH, 7.36 Âą 0.08. A pH level < 7.35 was found in 18.6%, and
PaCO2 >
45 in 57.4%. PaCO2 values
upon discharge were lower (46.1 Âą 4.6; p > 0.05). The ST (spontaneous-timed)
mode was used in 34 patients (79%). The ventilation period was 12.7 Âą 10.2
days, using 6.9 Âą 3.1 h/d. One third of patients received palliative care
(13.9% of mortality). Three patients (7%) were transferred to the Intensive
Care Unit due to clinical decline, and thirty-five were discharged with chronic
ventilation (94.6%).
Conclusions: there were few referrals to the Intensive Care Unit. Hospital mortality
was low, and patients who died had advance directives.
Key words: Non-invasive ventilation; Respiratory failure; Mortality.
RESUMEN
IntroducciÃģn:
La experiencia
clÃnica ha permitido la ventilaciÃģn no invasiva fuera de unidades crÃticas.
Describimos el perfil clÃnico y evoluciÃģn de pacientes que recibieron
ventilaciÃģn no invasiva en sala general.
Material
y mÃĐtodos: Estudio
retrospectivo en pacientes con soporte ventilatorio duÂrante un aÃąo en un
hospital general.
Resultados: Se utilizÃģ ventilaciÃģn no invasiva en 43 pacientes, 67,4% con hipercapÂnia. La relaciÃģn hombre/mujer fue 1:1. La edad y el IMC fueron 68,3 Âą 12,4 aÃąos y 30,1 Âą 12,3 kg/m2 y los diagnÃģsticos principales, enfermedad pulmonar obstructiva crÃģnica, enfermedad neuromuscular y obesidad-hipoventilaciÃģn. Un tercio iniciÃģ la ventilaciÃģn no invasiva en la unidad de cuidados intensivos, y dos tercios usaban ventilaciÃģn no invasiva en domicilio antes del ingreso por exacerbaciÃģn de la enfermedad pulmonar obstructiva crÃģnica (39,5%) o progresiÃģn de la enfermedad (14%). La estancia hospiÂtalaria fue 12,1 Âą 7 d (14 Âą 9 en supervivientes y 5,7 Âą 3 en pacientes fallecidos). La gasometrÃa arterial al ingreso revelÃģ PaCO2: 52,7 Âą 13,7 mmHg; PaO2: 72,2 Âą 16,2 mmHg y pH de 7,36 Âą 0,08. Se hallÃģ pH < 7,35 en el 18,6% y PaCO2 > 45 en el 57,4%. La PaCO2 al alta fue menor (46,1 Âą 4,6; p > 0,05). El modo ST se utilizÃģ en 34 (79%) pacientes. El perÃodo de ventilaciÃģn fue 12,7 Âą 10,2 dÃas con uso de 6,9 Âą 3,1 h/d. Un tercio recibiÃģ cuidados paliativos (13,9% de mortalidad). Tres pacientes (7%) fueron transferidos a la unidad de cuidados intensivos por deterioro clÃnico y treinta y cinco egresaron con ventilaciÃģn crÃģnica (94,6%).
Conclusiones:
Hubo
escasas transferencias a la unidad de cuidados intensivos. La mortalidad
hospitalaria fue baja y los que fallecieron tenÃan instrucciones anticipadas.
Palabras
clave: VentilaciÃģn
no invasiva; Insuficiencia respiratoria; Mortalidad.
Recibido: 01/08/2022
Aceptado: 08/05/2022
INTRODUCTION
The efficacy of non-invasive
ventilation (NIV) in respiratory failure was described in the â90s. From that
moment forward, it has been used to treat diseases that have traditionally been
managed at the Intensive Care Unit (ICU), such as chronic obstructive pulmonary
disease (COPD) and carÂdiogenic pulmonary edema.1
The use of NIV can reduce the need
for intuÂbation, shorten the hospital length of stay, and reduce mortality,
resulting in a rational use of the resources.2, 3 The acquired
experience has allowed its use outside the ICU.4
The failure or success can depend on the type of patient, the staffâs
skill, and follow-up tools. Each institution has to design its own response
protocol according to its resources.4-6
Frequently, patients with chronic
respiratory failure show exacerbations that require hospitalÂization.2-9 To decide
where to deliver NIV inside the hospital can be a complex decision involving
several factors such as the kind of underlying disease, associated
comorbidities, the severity of the physiological damage, and the patientâs
preferences.5-9
On the other hand, NIV is used in
environÂments without continuous monitoring, such as the patientâs home (HNIV),
where the ventilaÂtory treatment is guided by the
health care team and the family, for the purpose of improving the patientâs
quality of life.5-10
Finally, a group of patients with
progressive diseases or advance disease stage develop respiÂratory failure. In
these cases, NIV can be used in combination with other treatments to mitigate
dyspnea.10, 11
In 2017 we organized a
multidisciplinary team to deliver NIV at the regular hospital ward in patients
without immediate indication for ICU admission in the absence of clinical
severity signs (according to the Plant12criterion)
or advance diÂrectives for therapeutic limitation.
OBJECTIVES
To describe the clinical profile of
patients who received NIV in a regular hospital ward and their clinical
outcome.
MATERIALS AND METHODS
Retrospective study in patients
treated with NIV in a regular hospital ward.
The protocol was approved by the
Ethics and InstituÂtional Review Committee of the Hospital BritÃĄnico
de BueÂnos Aires, in accordance with the Declaration of Helsinki (protocol:
CRI# 1052, March 2020).
Population
The study included consecutive
adult patients who had been admitted to the regular ward of the Hospital BritÃĄnico de Buenos Aires between January and December 2019
(12 months), and received NIV. They were included when they were receiving HNIV
and were admitted due to an acute event, or if they were transferred from the
ICU to the regular ward. Patients with tracheostomy younger than 18 years were
excluded.
Clinical data
The following information was
obtained from the unique electronic medical record (EMR), SAPâĒ: medical
history, reeason for hospitalization, and previous
use of NIV. SpiÂrometries performed in our
institution (MedGraphics Paul. Saint
Paul, USA, Nhanes III reference equation) up to 3
months before were taken into account.
Our center is a clinico-surgical general hospital with 350 beds and 40 beds
for adult intensive care. The Respiratory Kinesiology, Pulmonology, Internal
Medicine and Intensive Care Departments have residents and personnel on duty 24
hours a day.
The indication and use of NIV was
decided by a multidisÂciplinary team. Patients were examined when they began
the NIV and two hours later, with daily periodic visits in the morning and at
night.
The ventilation parameters were
selected after the process of adaptation, gradually, taking into account the
clinical status until the patient achieved a balance between efficacy (objective)
and tolerance (comfort and compliance).
Ventilators were classified in
the following way:
â Basic, level I devices:
continuous flow generators for barometric ventilation with basic compliance
monitorÂing, without battery or alarms.
â Intermediate, level II devices:
continuous flow generaÂtors for barometric ventilation with battery and high
priority alarms, with ventilatory efficiency and
compliÂance monitoring.
â Advanced devices with life
support (level III): volume-controlled or pressure-controlled ventilation,
alarms of different priority levels, availability of internal and external
battery and full monitoring.
The ventilator choice was based
on clinical complexÂity (acute, chronic, palliative care), hours of use and
vital support requirement. The interface was selected for each patient. The
monitoring was obtained through a ventilator-integrated software; Encore Pro II
âĒ and Direct View âĒ (Philips-RespironicsâĒ. Murrysville USA) and ResScan 10.1âĒ (ResMedâĒ. San Diego, USA).
Follow-up included clinical
examination and physiologiÂcal parameters (vital signs, ventilatory
mechanics, state of consciousness, and pulse oximetry:
SatO2). The
basal arterial blood gas (ABG) was obtained in the morning. HyÂpercapnia
was defined as PaCO2 >
45 mmHg. The current protocol proposes that gasometry
should be performed daily in unstable patients during the first hospitalization
stage, and every 48 h or whenever there is clinical change during the patientâs
stay in the regular ward. We suggested daily data download from the internal
memory of the ventilators in accordance with our institutional protocol, with
review of efficiency and compliance and possible parameter adÂjustment. The
respiratory polygraphy was made whenever there was
inconsistent monitoring information, or in cases of lack of clinical
improvement after multiple parameter adjustments.
We reviewed the clinical outcome
(death, hospital disÂcharge, inclusion in a palliative care program, limitation
of therapeutic effort), hospitalization place, use of ventilatory
support, and referral to and from the ICU.
The decision to discharge
patients from the hospital was made jointly between participant services, once
the patient showed clinical stability, family and social support, organized
home care (in case this modality was applicable) and gasometric
improvement. Upon discharge, the patient coordinated daytime visits to hospital
and outpatient offices of the corresponding specialist physician. Figure 1 shows the current follow-up
protocol.
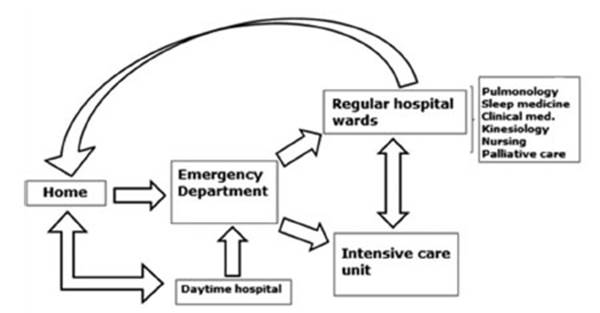
Statistical analysis
Categorical variables are
expressed in percentages.
In the case of continuous
variables with normal distriÂbution, results are expressed as mean and standard
deviaÂtion. Numerical variables without normal distribution are expressed as
median and percentile (25%-75%).
Differences between groups were
compared through the Mann-Whitney Test or the c2 Test for quantitative and
qualitative variables, respectively. Comparisons including three or more groups
were made through the Kruskal-Wallis Test and the
non-parametric Cochranâs Q Test.
A p value > 0.05 was
considered statistically significant.
The analysis was conducted with
the Prism 8.02 software (Graph Pad, La Jolla, CA).
RESULTS
NIV was delivered to 43 patients
of 68.3 Âą 12.4 years with a body mass index (BMI) of 30.1 Âą 12.3 kg/m2 for one year.
44.2% were obese and 67.4% showed hypercapnia. The
male/female ratio was 1: 1.
The diagnoses were (n =
%); COPD (17; 39.5%), neuromuscular diseases (16; 37.2%), obesity and
hypoventilation (5; 11.6%), heart failure (3; 7.0%) and thoracic cavity
restriction (2; 4.7%). Table 1 shows the characteristics of the population.
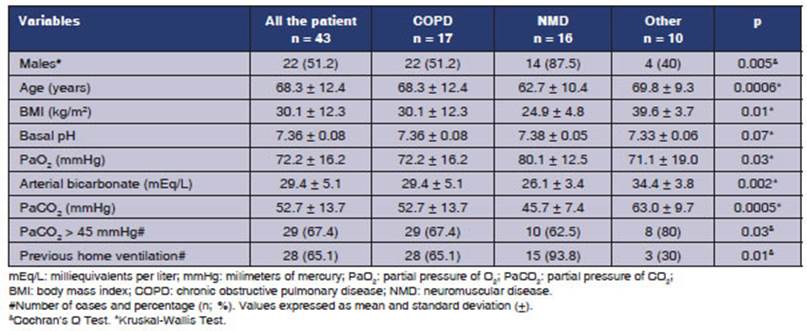
Reasons for admission were COPD
exacerbation (39.5%), heart failure (21%), progression
of muscle weakness (14%), pneumonia (11.6%), percutaneÂous gastrostomy (4.7%)
and emergency surgery (4.7%), among other things (4.7%). Hospital length of
stay was 12.1 Âą 7 d for all the population; 14 Âą 9 d for survivors; and 5.7 Âą 3
d for deceased patients.
Twenty-eight patients (65.1%)
were using HNIVat the time of admission. The remaining
15 patients (34.9%) began NIV at the ICU and were transferred to a regular ward
after they were stabilized.
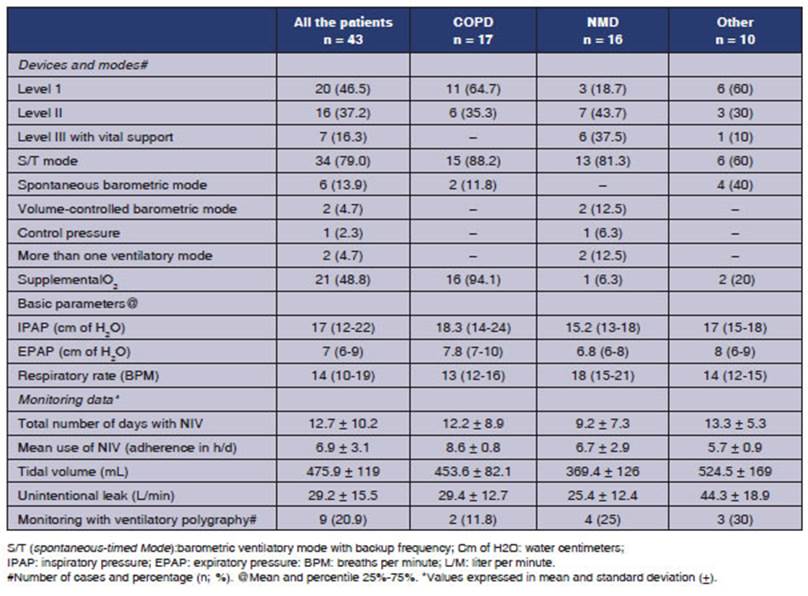
The barometric mode with backup
frequency (S/T) was the most widely used (79%) (Table 2).
Most patients (90%) used oronasal masks, and 21 (48.8%)
needed supplemental O2,
especially COPD patients (Table 2). The time NIV was use was 12.7 Âą 10.2 d,
with a compliance of 6.9 Âą 3.1 h/d, and we could observe more extensive use in
COPD (p < 0.01) (Table 2).
Twenty-nine patients had PaCO2 > 45 mmHg
upon admission (67.4%); and 8 had pH < 7.35 (18.6%).
Three patients (7%) were admitted
to the ICU due to clinical worsening (2 for impaired consciousÂness and one due
to progressive hypercapnia), though none of them
died.
Thirty-five patients were
discharged with HNIV (94.6% of dicharged subjects).
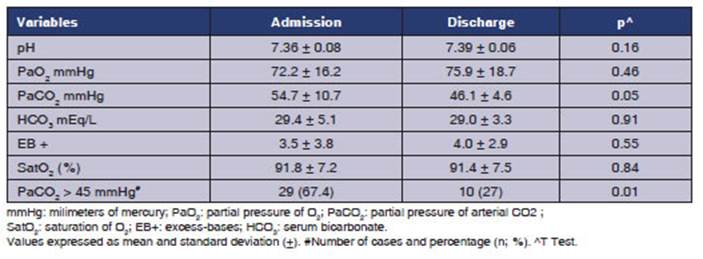
There were 7 (20%) new indications of home ventilation (Table 4). The PaCO2 upon
discharge was lower (46.1 Âą 4.6; p 0.05), even though 10 patients (27% of survivors)
were discharged with PaCO2 >
45 mmHg (Table 3).
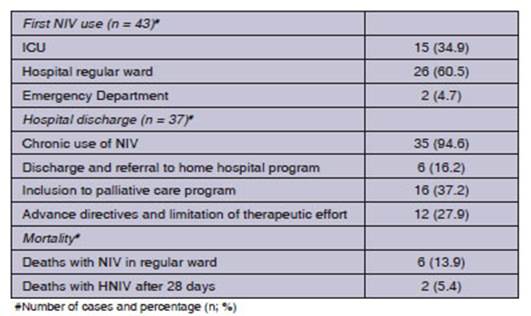
Finally, 16 patients (37.2%) were
included in palliative care programs (mortality of 37.5%: 6 patients); there
were 5 COPD patients and 5 cases of metastasised
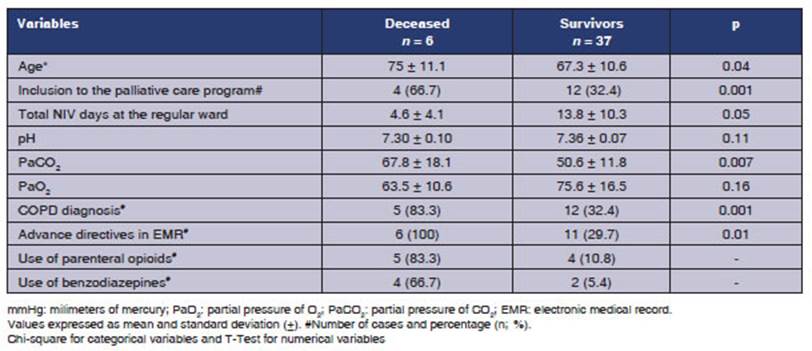
prostate cancer. The deaths were COPD-related and had some aspects in common:
patients were older and had advance directives for therapeutic limitation, high
PaCO2 levels
and took opioids to alleviate dyspnea or refractory symptoms (Table 5).
DISCUSSION
This analysis
describes the use of NIV in the regular ward of a general hospital in Argentina,
and shows that NIV was delivered to patients with COPD, neuromuscular disease,
hypoventilation due to obesity and respiratory failure (especially hypercapnic). A significant proportion of patients was
using HNIV or needed ventilatory support upon
discharge.
Some hospitals have
developed specialized reÂspiratory care units.13, 14 Our hospital doesnât have that type of unit, but we do treat
a considerable amount of NIV candidates outside de ICU. Even though general
ward rooms donât have centralized monitoring, we established the following
nurse/ patient ratio: 1:5, residents and kinesiologists
with active care shifts, 24 h a day.
The number of ICU
beds limits the number of patients who are admitted for respiratory failure. In
their study, Lapichino et al showed that there is a
tendency among intensive care physicians to give priority to surgical patients,
mostly those with clinical diseases,15 particulary
when unfaÂvorable results are expected. On the other hand, ICU admission of
less severe cases or patients with chronic diseases implies risks (for example,
infection, isolation, delirium, etc.) and increases healthcare costs.
NIV has been
delivered at the regular hospital ward for more than one decade. In an Italian
study, 56% of 756 patients were successfully treated with NIV (60% due to COPD
exacerbation). Also, 47% of the subjects were directly referred from the
emergency unit.16 In out center, patients
who had already adapted to NIV had priority for admission to a regular ward. A
small percentage of patients (7%) were transferred to the ICU.
In many parts of the
world,14 the regular ward environment is
considered inadequate for delivÂering NIV.4 However, a multicenter,
randomized, controlled trial,18 directed by Plant et al showed the
efficacy and safety of NIV in a regular ward in paÂtients with COPD
exacerbation and mild respiraÂtory acidosis (pH 7.30-7.35). When NIV is
delivered by qualified personnel, it reduces the number of occupied beds at the
ICU, the intubation rate and mortality.18 In
our series, due to preestablished safety criteria,
the admission pH (7.36) was higher than the value reported in similar studies.14-18
COPD exacerbation has
been the most analyzed disease for the use of NIV outside the ICU. It has been
suggested that a diagnosis other than COPD would be predictive of failure. More
experience is necessary so as to recommend NIV in a regular ward in other
situations.
COPD patients showed
high values of PaCO2 and hypoxemia and needed supplemental oxygen and
higher-pressure support, though they didnât reach the values suggested by some
authors.9 On the other hand, patients with NMD were younger and had
a much lower BMI. The fact that many of these patients had already received
HNIV can explain why this group showed closer to normal PaCO2 values.
Also, it was possible
to identify a heterogeneous group mainly composed of obese subjects with
alveolar hypoventilation, most of which hadnât been diagnosed before admission
and were characÂterized by high BMI, chronic respiratory acidosis (high level
of blood bicarbonate) and acidemia (exacerbation).
The 37 survivors with
NIV showed minimum pH deviation on admission (7.36 vs. 7.30) and a PaCO2 that
was lower than that of patients who died (51 mmHg vs. 68 mmHg), thus indicating
that the latter had severe respiratory failure. Besides, most patients in this
sample were using home NIV; this means they continued using a ventilatory support that was familiar to them. More than
80% of patients in this group showed severe acidemia
(pH < 7.35), so it would have been questionable to use limited resources
from the ICU environment. On this regard, it is interesting to see that the
existence of a non-invasive ventilation program avoided the use of valuable,
scarce, and expenÂsive resources (intensive care bed) for patients with HNIV,
many of which were hospitalized for intercurrent
conditions and required control and supplementary monitoring exams for the
purpose of optimizing the ventilatory support.
Almost half of the
patients used level I devices (basic). Vital support devices were used in NMD
patients who had greater dependence or needed to have multiple ventilation
modes.5 Even though the PaCO2 decreased with NIV and was close to
normalÂity upon discharge (46 mmHg), not every patient resolved hypercapnia (10 patients were discharged with PaCO2 > 45
mmHg). We must mention that our institutional protocol involves follow-up
through a daytime hospital model that prioritizes discharge with ventilatory support once the patient has adaptÂed to
ventilation and obtained basic comfortable parameters and improvement in pH and
PaCO2.
Our care model is
similar to the one used in North America and some European countries; the kinesiologist is included in the team that is responsible
for delivering NIV. Furthermore, the nursing staff can detect and resolve
problems and intolerance.19
According to the data
reported by developed countries, one fifth of NIV treatments begin in a regular
ward.20 However, logistic difficulties restrict the use of these
treatments. In Europe, Australia and New Zealand, shortage of personnel and inÂfrastructure
limit the use of NIV treatments.21-25 A
study of 157 centers in 51 countries of the five continents showed that 66% of
them use NIV outside the ICU. Inadequate training and limited human resources
are the reasons why NIV outside the ICU wasnât implemented.26
In Latin America,
data are limited. A survey conducted in fifteen hospitals in San Pablo, Brazil,
showed that private hospitals made greater use of NIV, where kinesiologists seemed to be more skilled (100%) than
physicians (73%) and nurses (33%).27
In Argentina,
information is scarce. According to Alonso et al, there are some areas where
NIV is not actively used28; and this could be considered an
indicator of poor sanitary quality. Also, the orÂganizational model of each
center determines the use of NIV outside the ICU.29-31 Our institution has a
daytime hospital where we begin HNIV;29
this can explain the differences with other series.15, 18-20
Many patients with
advanced cancer or progresÂsive diseases arenât candidates for endotracheal
intubation or invasive ventilation. A European study evaluated the
acceptability and efficacy of NIV vs. conventional oxygen therapy to reduce
dyspnea and the opioid dose. The results suggested that NIV is effective and
comfortable for patients with terminal cancer.10
In our series, two
thirds of patients with NIV and palliative care survived and were discharged.
Deaths occurred in patients with advance direcÂtives for limitation of
therapeutic effort. This finding shows the complexity of decisions at the end
of life in cases of respiratory failure, the difÂficulty to predict the outcome
and the role of NIV as âceiling of treatmentâ according to Roberts et al, in a
multicenter study of real-world.32
Azoulay et al conducted a prospective, multiÂcenter study on
the use of NIV in terminal paÂtients11 in 54 centers of France and
Belgium. 134 patients with a âdo not intubateâ order, survivors on day 90,
didnât show a reduction in their quality of life. In our series, 43% of
patients received NIV concurrently with opioids and anti-axiety
drugs. However, this scenario is limited to specialized centers after a
case-by-case discussion about the scope of treatment. In any case, NIV shall be
conÂtinued only if it is well-tolerated by the patient and if a benefit is
obtained. On the other hand, other measures must be taken to treat dyspnea (for
example, drugs). It is necessary to take into account the fact that in some
situations, NIV can unnecessarily prolong the life of the patient.33
This study has many
limitations, included those inherent to retrospective studies, and since it is
a single-center study with a heterogeneous populaÂtion, comparisons are
difficult to make. There isnât any control group, either, and most patients
with severe exacerbation criteria had advance directives of limitation of
therapeutic effort.
NIV was used in the
regular ward environment, especially in patients with COPD, neuromuscular
diseases and obesity with hypoventilation, and most of them had used some type
of ventilatory support before being admitted and were
discharged with indication of home ventilation. However, this perÂspective
enlightens us about the use of resources in this specific population in a
real-life scenario.
Both the number of
treatment failures requiring ICU admission and the inpatient mortality rate
were low. Deaths were recorded in patients with advance directives for
limitation of therapeutic effort.
Conflict of interest
Nothing
to declare.
Acknowledgement
To the regular ward
ventilation multidisciplinary team. Physicians:
Josefina Pascua, Pablo Lucero, BÃĄrbara Finn, Juan
Ignacio RamÃrez, Pablo Oyhamburu, Yael
GonzÃĄlez and Fernando Di Tullio. Respiratory
kinesiologists: Federico Melgarejo, Federico PÃĐrez,
Facundo Bianchini, Milagros Amedey,
Romina DomÃnguez, Emanuel Di Salvo, Alejandra Sosa, Mauro del Bono, Ignacio Brozzi and Josefina SÃĄez de Regadera. To the nurses of the regular ward who took care of
these patients.
REFERENCES
1. Hill NS, Garpestad
E, Schumaker G, Spoletini
G. NoninvaÂsive Ventilation for Acute Hypoxemic Respiratory Failure/ ARDS â is There a Role? Turk J Anaesthesiol Reanim 2017;45:332-4.
https://doi.org/10,5152/TJAR.2017,24,11,03
2. Lightowler
JV, Wedzicha JA, Elliott MW, Ram FS. Non-invasive
positive pressure ventilation to treat respiratory failure resulting from
exacerbations of chronic obstrucÂtive pulmonary disease: Cochrane systematic
review and meta-analysis. BMJ 2003;326:185.
https://doi.org/10,1136/bmj.326,7382,185
3. Keenan SP, Sinuff
T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic
obstructive pulmonary disease benefit from noninvasive positive-pressure ventiÂlation?
A systematic review of the literature. Ann Intern Med
2003;138:861-70.
https://doi.org/10,7326/0003-4819-138-11-200306030-00007
4. Farha
S, Ghamra ZW, Hoisington ER, Butler RS, Stoller JK. Use of noninvasive positive-pressure
ventilation on the regular hospital ward: experience and correlates of success.
Respir Care 2006;51:1237-43.
5. McKim
DA, Road J, Avendano M, et al; Canadian Thoracic
Society Home Mechanical Ventilation Committee. Home mechanical ventilation: a
Canadian Thoracic Society clinical practice guideline. Can Respir
J 2011;18:197-215. https://doi.org/10,1155/2011/139769
6. Hannan
LM, Dominelli GS, Chen YW, Darlene Reid W, Road J.
Systematic review of non-invasive positive pressure ventilation for chronic
respiratory failure. Respir Med 2014;108:229-43. https://doi.org/10,1016/j.
rmed.2013,11,010
7.
Masa JF, Corral J, Caballero C, Barrot E, et al. Non-invasive ventilation in obesity hypoventilation
syndrome without severe obstructive sleep apnoea. Thorax 2016;71: 899-906.
https://doi.org/10,1136/thoraxjnl-2016-208501
8. Struik
FM, Lacasse Y, Goldstein R, Kerstjens
HM, Wijkstra PJ. Nocturnal
non-invasive positive pressure ventilation for stable chronic obstructive
pulmonary disease. Cochrane Database Syst Rev
2013;2013:CD002878. https://doi.
org/10,1002/14651858.CD002878.pub2
9. KÃķhnlein
T, Windisch W, KÃķhler D, et
al. Non-invasive positive pressure ventilation for the treatment of severe
stable chronic obstructive pulmonary disease: a prospective, multicentre, randomised,
controlled clinical trial. Lancet Respir
Med 2014;2:698-705. https://doi.org/10,1016/S2213-2600(14)70153-5
10.
Nava S, Ferrer M, Esquinas A, et al. Palliative use
of non-inÂvasive ventilation in end-of-life patients with solid tumours: a randomised feasibility
trial. Lancet Oncol 2013;14:219-27.
https://doi.org/10,1016/S1470-2045(13)70009-3
11. Azoulay
E, Kouatchet A, Jaber S,
Lambert J, Meziani F, Schmidt M, et al. Noninvasive
mechanical ventilation in patients having declined tracheal intubation.
Intensive Care Med 2013;39:292-301.
https://doi.org/10,1007/s00134-012-2746-2
12. Plant PK, Owen JL, Elliott
MW. One year period prevalence study of respiratory acidosis in acute
exacerbations of COPD: implications for the provision of non-invasive venÂtilation
and oxygen administration. Thorax 2000;55:550-4. https://doi.org/10,1136/thorax.55,7.550
13.
Heili-Frades S, Carballosa
de Miguel MD, Naya Prieto A, et al. Cost and Mortality Analysis of an Intermediate ReÂspiratory Care Unit. Is It Really Efficient and Safe? Arch Bronconeumol
2019;55:634-41. https://doi.org/10,1016/j.arbr.2019,06,008
14.
Confalonieri M, Trevisan R,
Demsar M, et al. Opening of a respiratory intermediate care unit in a general hospital:
impact on mortality and other outcomes. Respiration 2015;90:235-42.
https://doi.org/10,1159/000433557
15.
Iapichino G, Corbella D, Minelli
C, et al. Reasons for refusal
of admission to intensive care and impact on mortality. InÂtensive Care Med 2010;36:1772-9.
https://doi.org/10,1007/s00134-010-1933-2
16. Confalonieri
M, Gorini M, Ambrosino N,
et al; Scientific Group on Respiratory Intensive Care of the Italian AssociaÂtion
of Hospital Pneumonologists. Respiratory intensive
care units in Italy: a national census and prospective coÂhort study. Thorax
2001;56:373-8. https://doi.org/10,1136/thorax.56,5.373
17. BarbÃĐ
F, Togores B, Rubà M, Pons
S, MaimÃģ A, Agustà AG.
Noninvasive ventilatory support does not facilitate
recovÂery from acute respiratory failure in chronic obstructive pulmonary
disease. Eur Respir J 1996;9:1240-5. https://doi.org/10,1183/09031936,96,09061240
18. Plant PK, Owen JL, Elliott
MW. Early use of non-invasive ventilation for acute exacerbations of chronic
obstructive pulmonary disease on general respiratory wards: a multiÂcentre randomised
controlled trial. Lancet 2000;355:1931- 5.
https://doi.org/10,1016/S0140-6736(00)02323-0
19. Hill NS. Where should noninvasive ventilation be delivered? Respir Care 2009;54:62-70.
20. Girou
E, Brun-Buisson C, TaillÃĐ
S, Lemaire F, BroÂchard L.
Secular trends in nosocomial infections and mortality associated with noninvasive
ventilation in patients with exacerbation of COPD and pulmonary edema. JAMA 2003;290:2985-91. https://doi.org/10,1001/jama.290,22,2985
https://doi.org/10,1001/jama.290,22,2985
21.
FernÃĄndez-Vivas M, GonzÃĄlez-DÃaz G, Caturla-Such J, et
al. UtilizaciÃģn de la ventilaciÃģn no invasiva en la insuficiencia respiratoria
aguda. Estudio multicÃĐntrico en unidades de cuidados
intensivos [Use of non-invasive ventilation
in acute respiratory failure. Multicenter study in intensive care units]. Med Intensiva 2009;33:153-60.
https://doi.org/10,1016/S0210-5691(09)71210-0
22. Ozsancak
Ugurlu A, Sidhom SS, Khodabandeh A, et al. Where is Noninvasive Ventilation
Actually Delivered for Acute Respiratory Failure? Lung 2015;193:779-88.
https://doi.org/10,1007/s00408-015-9766-y
23. Nasiłowski
J, Szkulmowski Z, Migdał
M, et al. RozÂpowszechnienie wentylacji
mechanicznej w warunkach domowych w Polsce [Prevalence of
home mechanical venÂtilation in Poland]. Pneumonol Alergol Pol 2010;78:392-8.
https://doi.org/10,5603/ARM.27695
24. Hazenberg
A, Cobben NA, Kampelmacher
MJ, Rischen J, Wijkstra PJ.
Chronische
beademing in Nederland [Home mechanical ventilation
in the Netherlands]. Ned Tijdschr Geneeskd 2012;156(3):A3609.
25. Garner DJ, Berlowitz DJ, Douglas J, et al. Home mechaniÂcal
ventilation in Australia and New Zealand. Eur Respir
J 2013;41:39-45.
https://doi.org/10,1183/09031936,00206311
26.
Cabrini L, Esquinas A, Pasin
L, et al. An international
survey on noninvasive ventilation use for acute respiraÂtory failure in general
non-monitored wards. Respir
Care 2015;60:586-92.
https://doi.org/10,4187/respcare.03593
27. NÃĄpolis
LM, Jeronimo LM, Baldini
DV, Machado MP, de Souza VA, Caruso P. Availability and use of noninvasive ventilation
in the intensive care units of public, private and teaching hospitals in the
greater metropolitan area of SÃĢo Paulo, Brazil. J Bras Pneumol 2006;32:29-34.
https://doi.org/10,1590/S1806-37132006000100008
28.
Alonso A, SchÃķnfeld D, Lopez
AM, Casas D, Violi JP, PenizÂzotto
M. Encuesta sobre el uso de VentilaciÃģn no invasiva en instituciones pÚblicas y
privadas Argentinas. Conociendo la realidad de su aplicaciÃģn. Rev Am Med Resp
2018;4:223-30.
29.
Blanco M, Ernst G, Di Tullio F, et al. Monitoreo de
la ventilaciÃģn domiciliaria crÃģnica. Experiencia basada en un modelo de
hospital de dÃa. Rev Am Med
Resp 2018; 3; 151-2.
30. Leske
V, Guerdile MJ, Gonzalez A, Testoni
F, Aguerre V. Feasibility of a pediatric long-term
Home Ventilation ProÂgram in Argentina: 11 yearsâ experience. Pediatr Pulmonol
2020;55:780-7. https://doi.org/10,1002/ppul.24662
31.
Borsini E, Blanco M, Ernst G, Ursino
R, Robaina G, Salvado A. Internaciones en pacientes con ventilaciÃģn
domiciliaria crÃģnica [Hospital admissions in patients with chronic
home ventilation]. Medicina (B Aires) 2018;78:403-9.
32. Roberts CM, Stone RA,
Buckingham RJ, et al; National Chronic Obstructive Pulmonary Disease Resources
and Outcomes Project implementation group. Acidosis, non-invasive ventilation
and mortality in hospitalised COPD exÂacerbations. Thorax 2011;66:43-8. https://doi.org/10,1136/ thx.2010,153114
33.
Tripodoro VA, Rabec CA, De
Vito EL. Withdrawing noninÂvasive ventilation at end-of-life
care: is there a right time? Curr Opin
Support Palliat Care 2019;13:344-50.
https://doi.org/10,1097/SPC.0000000000000471