Autor : Simonassi Julia InÃĐs1, Canzobre MarÃa Tatiana1
1Kinesiology Service, Hospital Nacional de PediatrÃa Juan P. Garrahan, Autonomous City of Buenos Aires, Argentina. .
https://orcid.org/0000-0001-5632-3018
Correspondencia : Simonassi Julia InÃĐs Servicio de kinesiologÃa. HosÂpital Nacional de pediatrÃa Juan P. Garrahan. Combate de los pozos 1881, CABA, Argentina. E-mail: jsimonassi@garrahan.gov.ar; juliasimonassi@gmail.com
ABSTRACT
Intrapulmonary
percussive ventilation (IPV) is a high-frequency mechanical bronchial hygiene
technique (MBHT) that favors secretion clearance and is considered an alternaÂtive
to the resolution of atelectasis.
This
is a prospective, observational and descriptive case series study conducted beÂtween
August 1st, 2019
and December 31st,
2019. The study included patients younger than 18 years on ventilatory support
who received at least one session of IPV in the intensive care unit.
The
primary objective of our study was to describe the characteristics of the
population in whom we used a home IPV device as MBHT in the PICU. On a
secondary level, we will describe the methodology for using this device and its
results.
Results:
18 patients were included; 48 IPV sessions were done. The main reason for doing
IPV was the atelectasis diagnosis (83.3%). The treatment was carried out both
in patients on IMV (invasive mechanical ventilation) (55.6%) and in patients
with non-invasive support (44.4%), whether it was NIMV (non-invasive mechanical
ventilation) or HFNC (high-flow nasal cannula). 53.3% of the patients showed
radiographic resolution of atelectasis, where 75% only required between one and
two sessions to resolve it. No severe complications were observed.
Conclusion:
This study allows us to describe the population receiving IPV and presents a
tool that could be useful for the resolution of atelectasis.
Key
words:
Intrapulmonary percussive ventilation, Pulmonary atelectasis, Pediatrics, Chest
physiotherapy, Mucus clearance
INTRODUCTION
The
pediatric patient shows certain anatomophysiÂological characteristics that
favor the development of respiratory complications, including narrowing of the
airways, low functional residual capacity and nondevelopment of collateral
ventilation1.
HospiÂtalization in the PICU (Pediatric Intensive Care Unit) and specially the
use of mechanical ventilaÂtion (MV) add several factors that somewhat favor
their appearance2, 3.
Also, analgesia and sedation used during ventilatory support have a relevant
role, because they alter the protective mechanisms of the airways4. On the
other hand, complications derived from the patientsâ stay at the PICU, as, for
example, acquired muscle weakness and loss of mobility, may cause retention of
secretions and subsequent development of atelectasis, worsening the situation
even more5.
Respiratory
physiotherapy (RP) is a set of techniques that contribute to mucociliary clearÂance,
favoring the removal of secretions retained in patients with difficulty in
managing secretions.6 Among these
techniques, there is the group of âhigh frequencyâ techniques, which are the
ones that generate high-frequency, low-volume oscillations during the
expiratory phase and can be produced both in an active and a passive manner.
These techniques can be used manually, through passive devices such as the
FlutterÂŪ or AcapellaÂŪ, or
mechanically, with the high-frequency chest compressor (VestÂŪ), the high-frequency chest oscillator
(The Hayek oscilÂlatorÂŪ)
and intrapulmonary percussive ventilaÂtion (IPV)7. The latter is a mechanical bronchial
hygiene technique (MBHT) where a high-pass, pulsatile flow is combined with low
tidal volumes delivered at high frequencies. This causes a transpulmonary
positive pressure gradient that favors alveolar recruitment and secretion clearÂance
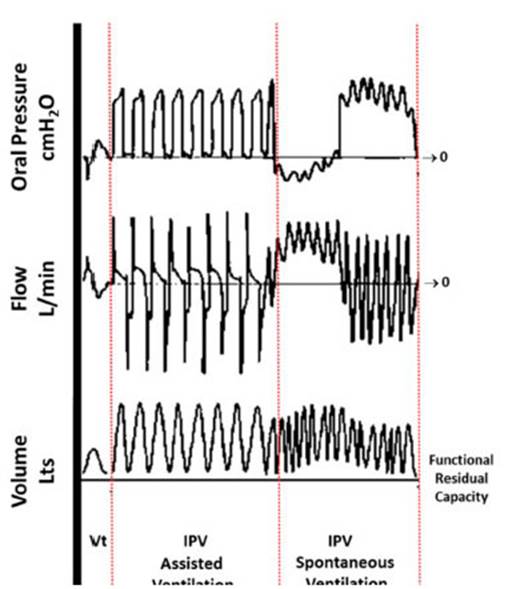
through an increase in the expiratory flow8
(Figure 1). In general, we can say there are two IPV devices
available on the market. Devices used in hospitals are pneumatic and can incorÂporate
high concentrations of oxygen (O2);
some are even combined with aerosol delivery and conÂtinuous airway pressure
(CPAP)9 whereas home
devices are electric and only deliver IPV without any other additional resources.
The frequency range used by these devices is 1.7-5 Hz, with pressures from 10
cmH2O up to
40 cmH2O; and
they are usually used in sessions that may last between 15 and 20 minutes10, 11.
They can be used with a nasal or oronasal mask, a mouthpiece, or directly
connected to the artificial airways with or without mechanical ventilation. the
greatest advantage of the IPV in pediatric patients is capacity to achieve
excellent coupling with sponÂtaneous ventilation; also, it doesnât need patient
cooperation and has very good tolerance7, 12, 13.
Several
authors showed the efficacy of the IPV in different populations of pediatric
patients14, 15 and explained
that the IPV is as effective as conventional bronchial hygiene techniques in
patients with cystic fibrosis16.
However, in pubÂlications related to the use of IPV in the PICU, we can see the
lack of evidence in this field. The only randomized clinical trial (RCT) is the
study in which it was proven that the IPV is a safe treatment and is effective
for the resolution of atelectasis in pediatric patients on mechanical
ventilation17.
Despite
the fact that evidence in favor of usÂing RP in the PICU is controversial in
certain scenarios, there are studies that support the use of IPV in critical
pediatric patients18,
19. However, the number of worldwide publications about
this topic so far is low and we couldnât find any studÂies conducted in Latin
America12, 17, 20.
Therefore, the primary objective of our study was to describe the
characteristics of the population in whom we used a home IPV device as MBHT in
the PICU. Secondary we will describe the methodology for using this device and
its results.
MATERIALS AND METHODS
This
study is a prospective, observational and descriptive case series conducted in
the Juan P. Garrahan pediatric hospital in the Autonomous City of Buenos Aires,
ArgenÂtina, in the period between August 1st, 2019 and December 31st, 2019.
The
study included patients younger than 18 years on ventilatory support who
received at least one session of IPV in the intensive care unit.
For
this study, an electronic record charts was designed with private access from
the mobile devices of the researchÂers. These were used to record demographic
variables, such as gender, age in months, weight, and also variables related to
the initial diagnosis, as for example, the presÂence of some kind of complex
chronic condition (CCC), the reason for using ventilatory support, whether it
was type 1 or type 2 acute respiratory failure (ARF) and the type of initial
ventilatory support21.
Also, IPV-related variables were recorded: indications for IPV (atelectasis,
hypersecreÂtion, hypoxemia), treatment duration, parameters of each session,
I/E (inspiration/expiration) ratio, pressure range, amount of cycles (number of
fractions of time within each session), in-line or independent use, and type of
ventilatory support required by the patient at the beginning of each session
(invasive mechanical ventilation, IMV; non-invasive mechanical ventilation,
NIMV; high-flow nasal cannula, HFNC, or extracorporeal membrane oxygenation,
ECMO). The use of additional O2
during treatment, complications, and clinical and result parameters
were recorded during each session.
The
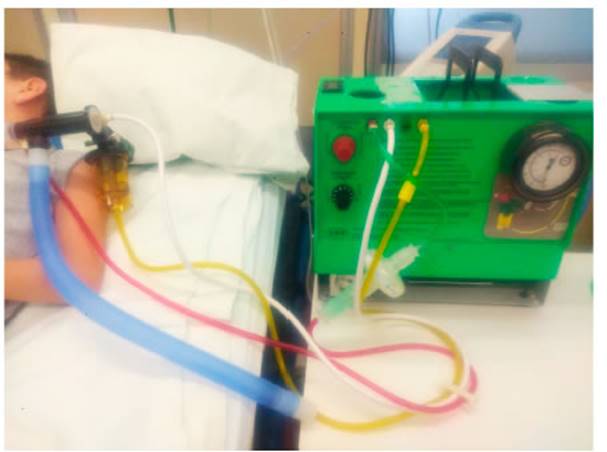
following home IPV devices were used for this study: The ImpulsatorÂŪ
from Percussionair (Sandpoint, Idaho, United States) (Figure 2) with
PhasitronÂŪ circuits (Sandpoint,
Idaho, United States). The parameters used at the discretion of the
physiotherapist according to the objective and tolerance of the patient were:
frequency of 90 cycles/min, 180 cycles/min and 250 cycles/min, I/E ratio of
1:1, 2:1 and 3:1 and a maximum pressure range of 10-40 cmH2O22,
23. In the cases where the device was used in-line, it was set in
assist pressure-controlled ventilation (PA/C-CMV) mode, with a positive
end-expiratory pressure (PEEP) of more than zero24. At the beginning of each sesÂsion, the
patient was placed lying on his/her back with a head elevation of 30°. In
patients with arterial O2 saturation
decrease below 88%, the corresponding cycle was suspended and continuation of
treatment was reconsidered according to clinical tolerance. If the event was
repeated, the session was suspended. As regards the duration of each session,
we considered a maximum of 20 minutes. In patients with atelectasis, the
frequency of the sessions was at least two per day, with one or two sessions in
the 8-16 h period and another one, according to the criterion of the treating
physiotherapist, in the 16-24 h period, during on-call time. In patients with
hypersecretion, the IPV was adapted to their bronchial hygiene plan, according
to the criterion of the health professional.
Due
to their asymmetrical distribution, continuous variables are expressed as
medians and interquartile ranges (IQR), and categorical variables are expressed
as frequenÂcies and percentages. For the data analysis, we used the IBM SPSSÂŪ Macintosh,
version 25.0 (IBM Corp., Armonk, NY, USA) statistical package.
Given
the fact that it is an observational study, informed consent wasnât required.
During the whole process, data were kept confidential and the identity of the
patients was preserved through numerical codification.
RESULTS
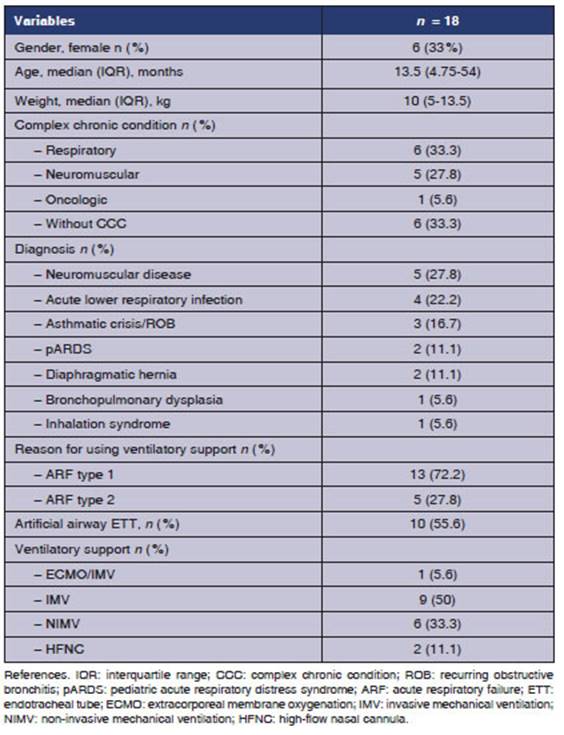
Eighteen
patients were included in the study; 33% of the patients were female, with a
median of age of 13.5 (4.75 - 54) months and a median of weight of 10 (5-13.5)
kg (Table 1).
The
66.7% of the patients had a CCC at study entry; the most common diagnosis was
neuromusÂcular disease (27.8%), and the reason for requiring ventilatory
support was type 1 acute respiratory failure (ARF) in 72.5% of the cases. A
total of 48 IPV sessions were carried out (Table 2).
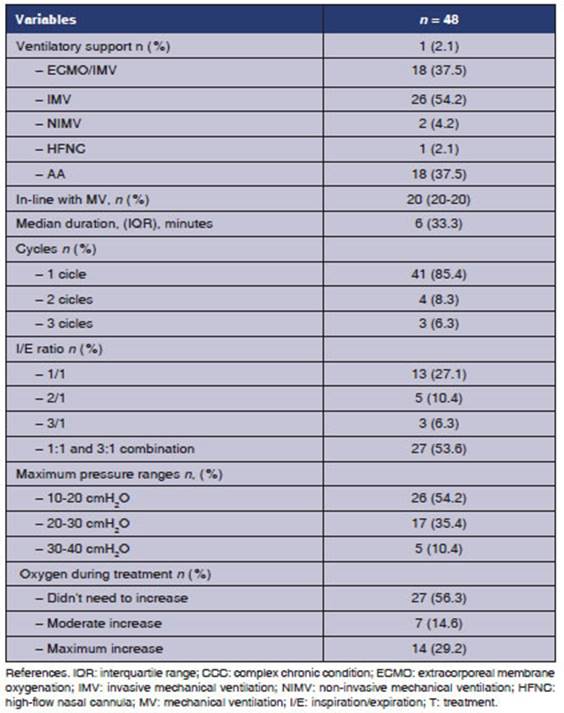
The
38.9% of the patients received one session; another 38.9% received two, and the
remaining 22.2% received three or more. The main reason for IPV use was the
atelectasis diagnosis (83.3%); other reasons were hypersecretion and hypoxÂemia.
The treatment was carried out both in patients who were on IMV (55.6%) and
patients with non-invasive support (44.4%), whether it was NIMV or HFNC. In
58.4% of the sessions, the patients were receiving some sort of non-invasive
ventilatory support. In 22.9% of the cases, the treatment was carried out
in-line with the ventilator. In the patient who was on ECMO, the IPV wasnât
in-line.
The
median duration of each session was 20 minutes. In 85.4% of the cases, it was
done in only one cycle without interruption. With regard to the way of using
the IPV, it was set with a 3:1 I/E ratio in 59.9% of the sessions; in 89.6% the
maximum pressure was lower than 30 cmH2O;
and in 43.7% of the sessions, it was necessary to increase the O2 during the
treatment. 80% of the patients who had sessions with a maximum pressure range
of 30-40 cmH2O were
on IMV with endotracheal tube.
The
53.3% of patients showed radiographic resolution of atelectasis, where 75% only
required between one and two sessions to resolve it. The saturation/fraction of
inspired O2 ratio
(SpO2/FiO2) improved in
14.6% of the sessions.
In
6.3% of the sessions, it wasnât possible to complete the prescribed time due to
desaturaÂtion. However, two out of three patients who had presented this
complication resolved the atelectasis.
DISCUSSION
This study allowed us to describe the population receiving IPV within
the environment of our inÂstitution, a reference center for Latin America. It
also showed us that it is possible to use home IPV devices in the PICU, since
they are effective with only a small number of sessions for the resolution of
atelectasis.
The demographic characteristics of our study were similar to those
of Morgan et al. mostly male patients, 63% of patients with a median of age of
2 years, and a median of weight of 14 kg20. The characteristics were
also similar to the Deakins group, which reported a median age of 3.1 years17.
The CCC of neurological origin is a characterÂistic that limits
the patientâs capacity to remove secretions effectively. In our study, the
group of patients with this condition required IPV just like all the patients included
in the study of Bidwala et al., who had fewer annual infections and used less
antibiotics and steroids13.
Birkrant et al. proposed that more research should be done for the
purpose of finding which are the diseases that would benefit most from the use
of this technique and describing more complications
during its implementation; accordingly, we provided a variety of diagnoses,
healthy patients and patients with CCC, patients with invasive and non-invasive
support and some minor complicaÂtions25.
The
major problem with respiratory physiotherÂapy in the pediatric population is
mainly the lack of cooperation and irritability to certain stimuli. For that
reason, the IPV could be presented as a good alternative to other techniques in
young chilÂdren, since it doesnât require patient cooperation. Individual setup
of percussion and frequency is generally well-tolerated, because it doesnât
require coordination with the patient; on the contrary, it is adjusted to the
patientâs respiratory rate17,
22, 25. It is even a good bronchial hygiene alternative in
patients of less than one year with gastroesophaÂgeal reflux, as shown in the
study of Lievens et al26.
In
most studies, there wasnât any adverse event12, 13, 16, 17, 27. Only Morgan and Homnick
deÂscribed an episode of pneumothorax and one mild hemoptysis, respectively20, 28.
Unlike those cases, we had three mild desaturation levels immediÂately solved
after treatment discontinuation. Some laboratory studies allow us to understand
that IPV would represent a low risk for barotrauma, in terms of the pressure
levels used, especially in pediatric patients on IMV29. Certain authors observed the pressure
drop phenomenon: with similar maximum inspiratory pressure compared to the
PA/C-CMV mode, the mean airway pressure in an IPV system is much lower. This
could be due to the shape of the pressure curve and the effect of percussions
on pulmonary geometry, which could produce a drop in the alveolar pressure30, 31.
Another relevant piece of information observed by Smallwood is that the
percentage of pressure reduction is inversely proportional to the size of the
tube, which reaches up to 60% in the 3.0 tube. This observation favors its use
in the neonatal and pediatric populations29.
According
to Deakins, the in-line use of IPV in patients who received invasive mechanical
ventilaÂtion is safe and effective for the resolution of atelÂectasis, just
like it was observed in our population. However, we believe our case series
study provides information mainly relating to the diversity of this MBHT in
patients with different clinical situations and ventilatory needs17.
In
the study of Rivera Cano et al., this technique was used as NIMV in patients
with bronchiolitis who didnât respond to CPAP, but it wasnât used as a MBHT.
Regarding the implementation of IPV in patients on ECMO, we could only find one
case report of a pediatric patient with Bordetella Pertussis who received IPV
to resolve a massive atÂelectasis, with good treatment response32, 33. There are studies where IPV was
used in patients with ARDS or ECMO as invasive ventilatory support, but not as
a MBHT, which is the object of study of this article34-37.
Pediatric
publications regarding the use of IPV as a MBHT in critical patients are
limited but promising, since they show the fact that it is a safe and effective
option for the resolution of atelectasis. The resolution rate of our study was
53.3%. The Deakins study used an atelectasis score that emphasizes a large
improvement in the IPV group compared to the group receiving conventional
respiratory physiotherapy, with a median of 3.1 days versus 6.2 days that the
control group took for the resolution3, 17. In our study, the group of
patients who resolved the atÂelectasis did it in a shorter period of time,
since they required only a median of two sessions. This difference could be due
to the fact that the duration of the sessions in our study was longer (10
minutes versus 20 minutes), and that, at the moment of the Deakins et al.
publication, there was no awareness about the way of optimizing the setup of
the IPV device24.
Yen Ha et al. could have provided additional data on this regard, but, the
methodology of radiographic diagnosis (all the patients underwent X-rays on day
2 and then on day 5) may have limited the findings, since there could have been
patients that immediately resolved the atelectasis but still continued with the
treatment for 5 days12.
The IPV is an effective MBHT that helps clear secretions and takes little time
to resolve atelectasis. Like other authors, we believe this characteristic
could be relevant due to the reduction in physiotherapy treatment time and
healthcare costs13,
17, 19.
With
respect to the duration of the IPV sesÂsions, we know there are two ways of
setting up the device that vary between 10 and 20 minutes. The reason for the
shorter duration could be asÂsociated with two situations: one is the fact that
there are some IPV devices such as the MetaNebÂŪ
System (Hillrom Services, Amsterdam, the NethÂerlands) that have
a 10-minute timer13,
20; and the other situation is that the session ends when
the nebulizer runs out of physiological solution, which lasts for approximately
10-15 minutes; that is the criterion for ending the session14, 17. Both in the work of Yen Ha et al.
and our work, the usage trend was between 15 and 20, as suggested by the Guides
published by the creator of this technology, Dr. Forrest Bird10, 12.
Some
authors describe the setup of the IPV, but generally not in a specific way. In
his in vitro study about the effects generated in the flow and pressure
by the different ways of setting up the device, Toussaint showed that the
expiratory flow increased with increasing I:E ratio.
This coincides with the I:E setup trend chosen by
physiotherapists in our hospital for 59.9% of the sessions12, 13, 22.
A
common characteristic in pediatric publicaÂtions about IPV was the small number
of patients. The study with the largest number of patients was the
retrospective descriptive analysis of Morgan et al., with 59 patients on IMV
and in-line use of IPV20;
in the RCT of Deakins et al., a total of 12 patients were admitted (5 in the
control group and 7 in the intervention group); Campbell Reardon et al.
included 18 neuromuscular patients; Yen Ha et al., 6 patients with atelectasis
and breathing difficulty; and Bidiwala et al., 8 tracheostomized chronically
critically ill patients12-14,
17. Our study with more than 18 patients provides
information about the versatility of the technique and treatÂment of atelectasis,
since it includes more cases than the rest of the PICU publications on this
topic.
LIMITATIONS
The
fact that we didnât use a score for the diagnosis of atelectasis could be
considered a bias; however, we chose not to use it because it is underused in
our work environment so, instead, we decided to use common radiographic
techniques for this purpose. Oxygenation through arterial gases could be an
outcome variable to consider in terms of the effiÂcacy of the technique; we
used SpO2/FiO2 to avoid
unnecessary invasive processes. The measurement of the volume of secretions was
dismissed, because there isnât any standardized method for this. Our work
doesnât allow the generalization of the reÂsults, but due to the
characteristics of the patients of our hospital, it wouldnât be completely
wrong to consider it as a useful tool to optimize secretion management in
patients who need it and to treat atelectasis in the PICU.
CONCLUSION
This
study describes the population in which IPV is population in which IPV is
implemented in the context of an institution in Argentina,
which is positioned as a benchmark in Latin America. It also presents a tool
that could be useful for the resolution of atelectasis within a short period of
time, optimizing its use in healthcare practice.
Conflicts
of interest
Authors
declare there isnât any conflict of interest or fiÂnancial support.
Acknowledgement
Gustavo
Plotnikow for his mentorship and a to critical care physiotherapy team of the
Juan P. Garrahan Hospital.
REFERENCES
1.
Oberwaldner B. Physiotherapy for airway clearance in paediatrics. Eur Resp J.
2000; 15: 196-204. https://doi.org/10.1183/09031936.00.15119600
2.
Walsh BK, Hood K, Merritt G. Pediatric Airway MainteÂnance and Clearance in the
Acute Care Setting: How To Stay Out of Trouble. Respiratory Care. 2011; 56:
1424-40. https://doi.org/10.4187/respcare.01323
3.
Lauwers E, Ides K, Van Hoorenbeeck K, Verhulst S. The effect of intrapulmonary
percussive ventilation in pediatric patients: A systematic review. Pediatric
Pulmonology. 2018; 53: 1463-74. https://doi.org/10.1002/ppul.24135
4.
Schechter MS. Airway Clearance Applications in Infants and Children. Resp Care.
2007;52:1382-90
5.
Chapman RL, Shannon H, Koutoumanou E, Main E. Effect of inspiratory rise time
on sputum movement during ventilator hyperinflation in a test lung model.
Physiotherapy. 2019; 105: 283-9. https://doi.org/10.1016/j.physio.2018.06.003
6.
Morrow BM. Chest Physiotherapy in the Pediatric IntenÂsive Care Unit. J
Pediatric Intensive care. 2015; 4: 174-81.
https://doi.org/10.1055/s-0035-1563385
7.
Chatburn RL. High-frequency assisted airway clearance. Respir Care. 2007; 52:
1224-35; discussion 1235-7
8.
Fernández-Carmona A, Olivencia-Peña L, Yuste-Ossorio ME,
Peñas-Maldonado L; Grupo de Trabajo de Unidad de Ventilación
Mecánica Domiciliaria de Granada. Ineffective cough and mechanical
mucociliary clearance techniques. Med Intensiva (Engl Ed). 2018; 42: 50-9. h
https://doi.org/10.1016/j.medine.2017.12.005
9.
Berlinski A, Willis JR. Albuterol delivery via intrapulmoÂnary percussive
ventilator and jet nebulizer in a pediatric ventilator model. Respir Care.
2010; 55: 1699-704.
10.
Percussionaire Corp. Clinical resources manual: IntrapulÂmonary percussive
ventilation (IPV). [Online]. [cited 2021 marzo 27. Available from:
https://issuu.com/percussionaire/docs/p20047_rev_a/24.
11.
Bird FM. Intrapulmonary percussive ventilation (IPV). Flying Physician. 1987;
30: 4-8
12.
Yen Ha TK, Bui TD, Tran AT, Badin P, Toussaint M, Nguyen AT. Atelectatic
children treated with intrapulmonary percussive ventilation via a face mask:
clinical trial and literature overview. Pediatr Int. 2007; 49(4): 502-7.
https://doi.org/10.1111/j.1442-200X.2007.02385.x
13.
Bidiwala A, Volpe L, Halaby C, Fazzari M, Valsamis C, Pirzada M. A comparison
of high frequency chest wall oscillation and intrapulmonary percussive
ventilation for airway clearance in pediatric patients with tracheostomy.
Postgrad Med. 2017; 129: 276-82. https://doi.org/10.1080/00325481.2017.1264854
14.
Reardon CC, Christiansen D, Barnett ED, Cabral HJ. IntraÂpulmonary percussive
ventilation vs incentive spirometry for children with neuromuscular disease.
Arch Pediatr Adolesc Med. 2005; 159(6): 526-31.
https://doi.org/10.1001/archpedi.159.6.526
15.
Toussaint M, De Win H, Steens M, Soudon P. Effect of intrapulmonary percussive
ventilation on mucus clearance in duchenne muscular dystrophy patients: a preliminary
report. Respir Care. 2003; 48: 940-7
16.
Natale JE, Pfeifle J, Homnick DN. Comparison of intraÂpulmonary percussive
ventilation and chest physiotherapy. A pilot study in patients with cystic
fibrosis. Chest. 1994; 105: 1789-93. https://doi.org/10.1378/chest.105.6.1789
17.
Deakins K, Chatburn RL. A comparison of intrapulmonary percussive ventilation
and conventional chest physiotheraÂpy for the treatment of atelectasis in the
pediatric patient. Respir Care. 2002; 47: 1162-7
18.
Johnston C, Zanetti NM, Comaru T, Ribeiro SN, Andrade LB, Santos SL. I
Brazilian guidelines for respiratory physiotherapy in pediatric and neonatal
intensive care units. Rev Bras Ter Intensiva. 2012; 24: 119-29.
https://doi.org/10.1590/S0103-507X2012000200005
19.
Van Ginderdeuren F, Vandenplas Y, Deneyer M, Vanlaethem S, Buyl R, Kerckhofs E.
Effectiveness of airway clearance techniques in children hospitalized with
acute bronchiolitis. Pediatr Pulmonol. 2017; 52: 225-31.
https://doi.org/10.1002/ppul.23495
20.
Morgan S, Hornik CP, Patel N, Williford WL, Turner DA, Cheifetz IM. Continuous
High-Frequency Oscillation Therapy in Invasively Ventilated Pediatric Subjects
in the Critical Care Setting. Respir Care. 2016; 61: 1451-5.
https://doi.org/10.4187/respcare.04368
21.
Feudtner C, Feinstein JA, Zhong W, Hall M, Dai D. Pediatric complex chronic
conditions classification system version 2: updated for ICD-10 and complex
medical technology dependence and transplantation. BMC Pediatr. 2014; 14: 199.
https://doi.org/10.1186/1471-2431-14-199
22.
Toussaint M, Guillet MC, Paternotte S, Soudon P, Haan J. Intrapulmonary effects
of setting parameters in porÂtable intrapulmonary percussive ventilation
devices. Respir Care. 2012; 57: 735-42. https://doi.org/10.4187/respcare.01441
23.
Riffard G, Toussaint M. Indications de la ventilation à percussions
intrapulmonaires (VPI): revue de la litÂtérature [Indications for
intrapulmonary percussive ventilation (IPV): a review of the literature]. Rev
Mal Respir. 2012; 29: 178-90. https://doi.org/10.1016/j.rmr.2011.12.005
24.
Riffard G, Buzenet J, Guérin C. Intrapulmonary percusÂsive ventilation
superimposed on conventional mechanical ventilation: comparison of volume
controlled and pressure controlled modes. Respir Care. 2014; 59: 1116-22. https://doi.org/10.4187/respcare.02727
25.
Birnkrant DJ, Pope JF, Lewarski J, Stegmaier J, BesunÂder JB. Persistent
pulmonary consolidation treated with intrapulmonary percussive ventilation: a
preliminary report. Pediatr Pulmonol. 1996; 21: 246-9. https://doi.org/10.1002/(SICI)1099-0496(199604)21:4<246::AID-PPUL8>3.0.CO;2-M
26.
Lievens L, Vandenplas Y, Vanlaethem S, Van Ginderdeuren F. The influence of
Intrapulmonary percussive ventilation on gastroesophageal reflux in infants
under the age of 1 year. Pediatr Pulmonol. 2020; 55: 3139-44.
https://doi.org/10.1002/ppul.25045
27.
Newhouse PA, White F, Marks JH, Homnick DN. The intrapulmonary percussive
ventilator and flutter device compared to standard chest physiotherapy in
patients with cystic fibrosis. Clin Pediatr (Phila). 1998; 37: 427-32.
https://doi.org/10.1177/000992289803700705
28.
Homnick DN, White F, de Castro C. Comparison of effects of an intrapulmonary
percussive ventilator to standard aeroÂsol and chest physiotherapy in treatment
of cystic fibrosis. Pediatr Pulmonol. 1995; 20: 50-5.
https://doi.org/10.1002/ppul.1950200110
29.
Smallwood CD, Bullock KJ, Gouldstone A. Pressure atÂtenuation during
high-frequency airway clearance therapy across different size endotracheal
tubes: An in vitro study. J Crit Care. 2016; 34: 142-5.
https://doi.org/10.1016/j.jcrc.2016.03.004
30.
Dutta R, Xing T, Swanson C, Heltborg J, Murdoch GK. ComÂparison of flow and gas
washout characteristics between pressure control and high-frequency percussive
ventilation using a test lung. Physiol Meas. 2018; 39: 035001.
https://doi.org/10.1088/1361-6579/aaaaa2
31.
Rožánek M, Horáková Z, Cadek O, Kučera M,
Roubík K. Damping of the dynamic pressure amplitude in the ventilatory
circuit during high-frequency oscillatory venÂtilation. Biomed Tech (Berl).
2012; 57:Suppl 1.
https://doi.org/10.1515/bmt-2012-4481
32.
Ribera Cano A, Daussac E, Bonnet S, Marcoux MO, Lelong- Tissier MC. Ventilation
non invasive par percussion intra pulmonaire (IPV) dans les
broncho-alvéolites virales [Non invasive intrapulmonary percussive
ventilation (IPV) in viral bronchiolitis]. Arch Pediatr. 2009; 16: 732-4.
https://doi.org/10.1016/S0929-693X(09)74130-2
33.
Krawiec C, Ballinger K, Halstead ES. Intrapulmonary Percussive Ventilation as
an Airway Clearance Technique during Venoarterial Extracorporeal Life Support
in an Infant with Pertussis. Front Pediatr. 2017; 5: 99.
https://doi.org/10.3389/fped.2017.00099
34.
Tawfik DS, Bennett TD, Welch B, Poss WB. Use of High- Frequency Ventilation in
the Pediatric Intensive Care Unit. J Pediatr Intensive Care. 2016; 5: 12-20
35.
Rizkalla NA, Dominick CL, Fitzgerald JC, Thomas NJ, Yehya N. High-frequency
percussive ventilation improves oxygenation and ventilation in pediatric
patients with acute respiratory failure. J Crit Care. 2014; 29: 314.e1-7.
https://doi.org/10.1016/j.jcrc.2013.11.009
36.
Yehya N, Dominick CL, Connelly JT, Davis DH, Minneci PC, Deans KJ, McCloskey
JJ, Kilbaugh TJ. High-frequency percussive ventilation and bronchoscopy during
extracorÂporeal life support in children. ASAIO J. 2014; 60: 424-8.
https://doi.org/10.1097/MAT.0000000000000088
37.
Butler AD, Dominick CL, Yehya N. High frequency percusÂsive ventilation in
pediatric acute respiratory failure. Pediatr Pulmonol. 2021; 56: 502-8
https://doi.org/10.1002/ppul.25191