Autor : Zurita Ingrid1, González Alejandra1, Idoyaga Pablo1Â
1 Department of Respiratory Medicine. Hospital Nacional Prof. Dr. Alejandro Posadas. El Palomar. Province of Buenos Aires. Argentina
Correspondencia : Dra. Ingrid M. Zurita Villarroel Ramos Mejia CP 1704. Ciudad AutĂłnoma de Buenos Aires Argentina E-mail: ingridzurita87@gmail.com
ABSTRACT
Paciente
de sexo masculino, 70 años, con leucemia mieloide crónica en
tratamiento con dasatinib, desarrolla insuficiencia respiratoria asociada a
toxicidad pulmonar por dicho fármaco.
Key
words:
Dasatinib; Tyrosine Kinase Inhibitor; Pulmonary Toxicity
RESUMEN
Paciente
de sexo masculino, 70 años, con leucemia mieloide crónica en
tratamiento con dasatinib, desarrolla insuficiencia respiratoria asociada a
toxicidad pulmonar por dicho fármaco.
Palabras
clave:
Dasatinib; Inhibidor de la tirosina quinasa; Toxicidad pulmonar
Received: 03/14/2021
Accepted: 07/20/2021
INTRODUCTION
Patients
with lymphohematological neoplasia may develop pulmonary diseases as a
consequence of the immunosuppression that is typical of the disease or the one
produced by the chemotheraÂpeutic or immunomodulating agents used for the
treatment1.
There
is a wide variety of drugs available for the treatment of this condition.
Various regimens that have shown potential pulmonary toxicity are of particular
interest to the pulmonologist who has to deal with differential diagnoses2.
CASE REPORT
70-year-old
male patient with the following medical history: bilateral blindness, renal
carcinoma with nephrectomy plus radiation therapy, and chronic myeloid
leukemia. The patient received treatment with imatinib for one year, and then
switched to dasatinib, 140 mg/d due to therapeutic failure.
The
patient went to the emergency service (before the pandemic) after 15 days of
dry cough, fever and progressive dyspnea. Chest X-ray was performed: right lung
alveolar infiltrate. A blood culture was taken and the patient began treatÂment
with levofloxacin, 750 mg/d with diagnosis of pneumonia. After 72 h, due to a
lack of clinical response, it was decided that the patient had to be
hospitalized. On admission, the patient had a fair general condition, with
fever, saturating 93% on room air. Laboratory tests: Hb (hemoglobin): 8 g/dL;
PCR (polymerase chain reaction) 9 mg/L; no leukocytosis; creatinine of 1.6 mg/dL.
New blood cultures are taken and the patient begins antibiotic treatment with
ampicillin/sulbactam + clarithromycin by endovenous route.
Progressive
deterioration, tachypnea and use of mask with reservoir bag due to respiratory
failure. Treatment switch to meropenem and transfer to intensive care unit due
to persistent fever. Chest tomography: air trapping, bilateral apical alveolar
infiltrate, bilateral pseudonodular images, mild left pleural effusion (Figure
1).
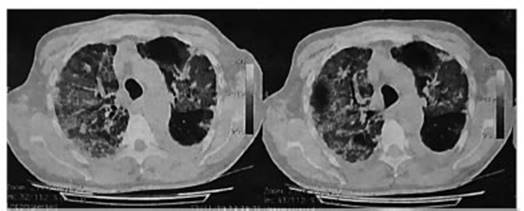
Normal
echocardiogram. Fibrobronchoscopy with bronchoalveolar lavage performed.
Culture for common germs, microbacteria and fungi: negative. PCR for Pneumocystis
jiroveci: negative. Negative cytological test.
Dasatinib
was suspended under suspicion of pulmonary toxicity, and replaced with corticoÂsteroid
treatment showing clinical, gasometric and imaging improvement. Patient is
discharged after 7 days, with indication for one month of oral corticosteroids.
New outpatient chest tomography without pulmonary infiltrates required (Figure
2).
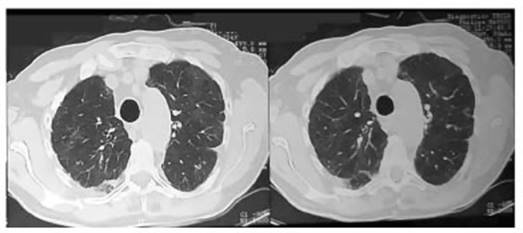
Dasatinib
is a second-line ABL tyrosine kinase inhibitor, with oral bioavailability,
active against BCR-ABL mutants resistant to imatinib. It is also used as a
first-line treatment.3, 4 The adverse
events include fever, myalgia, pulmonary arterial hyperÂtension, pleural
effusion (exudate), bronchospasm and pulmonary infiltrates (“ground glass”,
septal thickening, focal or pseudonodular consolidation). Pulmonary
manifestations normally appear one month after first day of treatment and can
appear up to two years post-treatment. They seem to have a dose-dependent
effect, probably the result of an immune-mediated mechanism5, 6.
The
Bergeron study identified 9 out of 40 patients (22.5%) with chronic myeloid
leukemia (CML) in chronic phase who received dasatinib and develÂoped pulmonary
anomalies. Six of those patients had pleural effusion (all exudated), and seven
subjects showed changes in the lung parenchyma with “ground glass” or alveolar
opacities and septal thickening (four patients had pleural effusion and changes
in the pulmonary parenchyma)7.
With
regard to the pleural effusion, if it has the necessary volume, a diagnostic
thoracocentesis must be performed to differentiate the exudate from the
transudate and also a culture has to be done to discard pleural infection. In
cases of minimum liquid, continuation of the tyrosine kinase inhibitor (TKI)
may be considered, with close clinical and radiological monitoring, whereas in
cases of moderate or large pleural effusion, it would be suitable to consider
dose reduction, TKI suspension or switch to an alternative treatment option for
CML8.
The
thoracocentesis, bronchoalveolar lavage and biopsy show lymphocyte predominance
(with lymphatic build-up shown in the biopsy)9.
Drug
suspension and subsequent use of systemic corticosteroids resolved the
condition. The drug can be used again in the minimum effective dose without
worsening the symptoms10.
CONCLUSION
Dasatinib-related
pulmonary toxicity shall be included as one of the differential diagnoses of
patients with respiratory symptoms and pulmonary infiltrates. Lack of response
to antibiotics, the negative result of bacteriological tests, and the clinical
and imaging progression led to the suspicion of the entity. The improvement
observed after drug suspension and the use of corticosteroids confirmed the
diagnosis.
Conflict
of interests
Authors
declare there isn’t any conflict of interest in relaÂtion to the contents of
this article.
REFERENCES
1.
Shorr AF, Susla GM, O’Grady NP. Pulmonary Infiltrates in the Non-HIVInfected
Immunocompromised Patient Etiologies, Diagnostic Strategies, and Outcomes.
Chest. 2004; 125; 260-71.
2.
Crawford SW. Noninfectious lung disease in the immunoÂcompromised host.
Respiration. 1999;66:385-95. https://doi.org/10.1159/000029418
3.
Kantarjian HM, Talpaz M, O’Brien SG, et al. High dose imatinib mesylate therapy
in newly diagnosed Philadelphia chromosome positive chronic phase myeloid
leukaemia. Blood 2004;103:2873-8.
4.
O’Brien SG, Guilhot F, Larson RA, et al. Imatinib comÂpared with interferon and
low dose cytarabine for newly diagnosed chronic phase chronic myeloid
leukaemia. N Eng J Med 2003;348:994-1004.
https://doi.org/10.1056/NEJMoa022457
5.
Breccia M, Latagliata R, Stagno F, et al. Charlson comorbidÂity index andadult
comorbidity evaluation-27 scores might predict treatment compliance and
development of pleural effuÂsions in elderly patients with chronic
myeloidleukemia treated with second-line dasatinib. Haematologica. 2011;96:1457-61.
6.
Valent P. Severe adverse events associated with the use of second-line BCR/ABL
tyrosine kinase inhibitors: PreferÂential occurrence in patients with
comorbidities. HaemaÂtologica. 2011;96:1395-7.
7.
Bergeron A, Réa D, Levy V, et al. Lung abnormalities after dasatinib
treatment for chronic myeloid leukemia: a case series. Am J Respir Crit Care
Med. 2007;176:814-8. https://doi.org/10.1164/rccm.200705-715CR
8.
Weatherald J, Bondeelle L, Chaumais MC, et al. PulÂmonary complications of
Bcr-Abl tyrosine kinase inÂhibitors. Eur Respir J. 2020;56:2000279.
https://doi.org/10.1183/13993003.00279-2020
9.
Rousselot P, Bergeron A, Re´a D, et al. Pleural and pulmoÂnary events in
patients treated with dasatinib for chronic myeloid leukemia in chronic phase
[abstract]. HaematoÂlogica 2007;92:A546.
10.
Grimison P, Goldstein D, Schneeweiss J, Murray N. Corticosteroid-responsive interstitial
neumonitis related to imatinib mesylate with successful rechallenge, and poÂtential
causative mechanism. Intern Med J 2005;35:136-7.
https://doi.org/10.1111/j.1445-5994.2004.00757.x