Autor : Jimena Falco, Vanina Martin, Leandro Marozzi, Marco A. SolĂs Aramayo, Marcos Hernández, Edgardo Sobrino, Silvia Quadrelli
Servicio de Neumonologia, Sanatorio GĂĽeme
Correspondencia : Falco Jimena L. DirecciĂłn: Caseros 275, Ezeiza, CP: 1804 e-mail: falcojimena@hotmail.com Tel: 1161749651/ 42329630
Abstract
The chronic obstructive pulmonary disease (COPD) is characterized by persistent airflow
limitation. It is a frequent, known and treatable disease. The diagnosis requires tobacco
smoking personal history or expositional history and evidence of an obstructive pattern
at the spirometry measured before and after bronchodilators.
The degree of reversibility of airflow differs between patients, and does not predict the
response to bronchodilator therapy. It can be seen in terms of forced vital capacity or
forced expiratory flow, depending on the severity of the disease.
This paper presents the differences in bronchodilator response in terms of measurements of
the flow and the volume in patients diagnosed with COPD at different stages of GOLD. The
bronchodilator response and its relationship to the severity of the disease were analized.
Key words: COPD; Spirometry; Vital capacity; Forced expiratory volume
Introduction
According to ATS/ERS task force, bronchodilator
response is measured using the percent change
from baseline and absolute changes in FEV1 and/or FVC1. Twelve percent and 200 mL increase in
FEV1 or FVC compared to baseline value are suggestive of a significant response to bronchodilator1, 2. However, some patients with severe chronic
obstructive pulmonary disease (COPD), forced
vital capacity (FVC) remarkably increases in response to bronchodilator administration whereas
FEV1 remains substantially unchanged3-5. This
isolated volume response is generally interpreted
as sign of bronchodilatation6. The reductionof
hyperinflation7 and/or the presence of severe
emphysema8 have been proposed as underlying
mechanisms of this volume response.
If severe emphysema and lung inflation alters
airway caliber, due either to loss of lung elastic
recoil or to space competition, airway calibre
should be independent of airways smooth muscle
tone at high lung volume, explaining at least in
part the lack of sensitivity to bronchodilatation
as assessed by changes in FEV18. It would mean
that the isolated volume response should be more
prevalent in more severe disease. The objective of our study was to assess the differences between
flow and volume responses after bronchodilator reversibility testing in patients over different clinical
COPD stages (GOLD stage I to GOLD stage IV) in
order to test if the association between flow and
volume responses becomes weaker as the disease
becomes more severe
Material and methods
We retrospectively reviewed the pulmonary
function of 594 consecutive patients with chronic
obstructive pulmonary disease who attended our
laboratory between January 2013 and June 2014.
Patients were included only if the referral diagnostic of the pulmonologist in charge was COPD and
were 40 years of age or older and the spirometry
confirmed a post-bronchodilator FEV1/FVC ratio < 0.70. When two or more tests of the same patient
were included in the database, the most recent
test for each individual patient was selected. All
patients who reported to be lifelong non-smokers
were excluded. Any patients with a diagnosis of
bronchial asthma, bronchiectasis or any other
respiratory disease were excluded.
All spirometric measurements were made using
identical spirometers, pneumotrac 6800 vitalograph with pneumotachograph of Fleisch and
software Spirotracacoording to standardisation
of spirometry ATS/ERS9.
Forced expiratory manoeuvres were performed
in a standardised fashion and the best FEV1 and
FVC recordings within 50 ml of each other were
accepted. All studies were performed by the same
3 technicians that met proficiency requirements
in the use of the equipment and demonstrated
the ability to perform technically acceptable pulmonary function tests according to ATS criteria.
Patients were asked to omit short-acting inhaled
bronchodilators for at least 8 hours, and longacting beta-agonists for at least 12 hours. Bronchodilator responsiveness was assessed by performing
spirometry before and 15 minutes after the patient
had received salbutamol, 400 μg, inhaled using a
spacer in 4 separate doses). Bronchodilator response is expressed as an absolute change in millilitres
and as a percentage of the baseline value.
FEV1 responsiveness was assessed using published criteria: >12% and >200 mL improvement10, 11 and also an increase in FVC of at least 200 mL and
12% from baseline values, we used these criteria
in our study10.
Patients were stratified according to the severity
of obstruction into one of the four GOLD stages:
mild (stage I: FEV1 > 80% of predicted); moderate (stage II: FEV1 50-80% of predicted); severe
(stage III: FEV1 30-50% of predicted); and very
severe COPD (stage IV: FEV1 o 30% of predicted)12.
Information regarding the presence of chronic
respiratoryfailure was not available, therefore the
criterion FEV1 < 50% of predictedplus chronic
respiratory failure to classify a patient in GOLD
stage IV was not applied.
Statistical analysis:
Data are presented as mean plus standard deviation for whole group data. Comparisons were made
using analysis of variance (ANOVA) for multiple
groups. Bonferroni test showed: A significant difference for all the included variables in the Group
of volume responders compared to flow responders and non responders. However the group of
non-responders was not different from the flow
responders. Differences in subject characteristics
between GOLD stage subgroups were analysed
by analysis of variance (ANOVA). Differences in
flow and volume responses between GOLD stages
and interaction between flow response, volume response, and GOLD stage were statistically tested
using a linear regression model with deltaFVC as
the dependent variable. Statistical analysis was
performed using SPSS version 13.0 and significance set at a level of 95% (p < 0.05).
Results
After applying our selection requirements, 594 patients fulfilled the entry criteria and had available complete data of bronchodilator assessments. The overall proportions of males in this sample was 66, 5% (n = 395). The age range in the sample was 40–93 years. Baseline lung function of the whole population isshown in Table 1. Neither mean age (p =0,464) nor mean BMI (p= 0,308) were different amongst the patients with different GOLD stages. Most of patients were classified as GOLD II (Table 2).
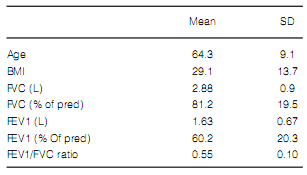

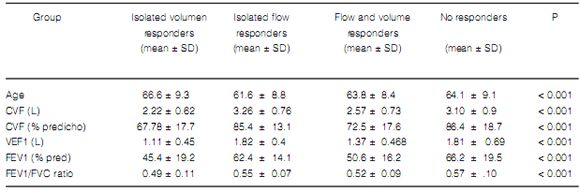
Table 4 shows the responses on flow and volume after the administration of bronchodilators in the different GOLD stages. The mean values for the change in FEV1 (DFEV1) and in FVC (DFVC) were 0, 15 ± 0, 13 L and 0, 21 ± 0, 26 Lrespectively . The magnitude of DFEV1 decreased as the GOLD stage became more severe and the DFVC increased in the more severe GOLD stages.

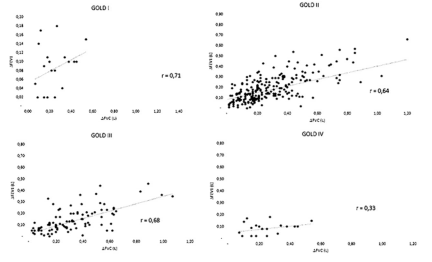
There was a positive correlation between DFEV1 and DFVC within the GOLD stages I to III (GOLD I: r = 0, 71, p = < 0,001; GOLD II r = 0, 64, p = < 0,001, GOLD III: r = 0,689, p = < 0,001) but not in GOLD stage IV (r = 0, 33, p = 0,135) (Fig 1).
Discussion
Our study demonstrated that a considerable
amount of COPD patients show some sort of
response to bronchodilators and that the more
severe the airway obstruction the higher the
probability of showing only volume response to
bronchodilators.
Bronchial responsiveness is usually assessed by
the change in the FEV1 after the administration of
a bronchodilator. Other measures of lung function,
such as lung volumes, are not routinely assessed
before and after the administration of a bronchodilator agent in most pulmonary laboratories. The
proportion of patients classified as “responders” will entirely depend on the definition of responsiveness. For instance, in the cohort studied by
Tashkin et al, 65.6% met the criterion of a o15%
increase in FEV1; 53.9% met the criterion of an
increase in FEV1 of both o12% and o200 mL (73%
of patients showed an increase of o12%, and55%
of o200 mL); and 38.6% were characterised asreversible based on a o10% absolute improvement
inpercentage predicted FEV113. By using the
currently ATS/ERS criteria, the magnitude of
bronchodilator responsiveness was greater than
expected with more than 40% of patients showing
some type of responsiveness. Until recently, COPD
had been characterised as a disease with largely
irreversible airflow obstruction. Although it is
now widely accepted that COPD is characterised
by partially reversible airflow obstruction, the
magnitude of acute responsiveness to bronchodilator drugs for COPD has not been rigorously
analysed13. In fact, the distinction from chronic asthma with limited reversibility remains difficult,
and many physicians still use in their daily practice
the spirometric response to a bronchodilator drug
to aid the diagnosis and, in some cases, to make
recommendations about treatment decisions. In
spite of the fact that some previous studies have
found no clear distinction spirometrically between
asthma and COPD in milder disease14-16.These
criteria have been still applied in the selection of
patients for inclusion in treatment trials whose
results are still in use17, 18.
There was a heterogeneous response in our population with 79 (13, 3%) subjects showing isolated
volume response, slightly more patients (n = 118,
19.9%) exhibiting changes in both volume and flow
and only 5.1% improving FEV1 with a little change
in FVC. This latter group had better initial spirometry. This distribution shows a slightly lower
prevalence of responders than the series of Walker&Calverley in266 COPD patients who attended
theirpulmonarylaboratory in Liverpool. Those
differences may be explained because of a different
definition of responsiveness, as the subjects in
their study were classified as volume responders if
FVC increased > 330 ml post-bronchodilator and
FEV1 by < 160 ml19. In accordance to our findings,
their patients who were in the volume responder
group had a significantly lower baseline FEV1.
They also demonstrated that this group exhibited
a greater reduction in inspiratory capacity and an
increased residual volume compared to the unresponsive and flow responsive patients alone. In our patients in the volume responder group, the FEV1/FVC ratio tended to worsen after the bronchodilator, probably due to the disproportionate increase
in volume relative to flow.
The change in FEV1, expressed as an absolute
value was uninfluenced by the pre-bronchodilator
FEV1 when measured in absolute units. However,
when the data were expressed as a percentage
change from baseline there was a clear influence
of the pre-bronchodilator FEV1; the more severe
the baseline obstruction, the higher the increase
of FEV1 as a percent of the baseline value. This
has also been shown in the Calverley’ study in
patients recruited from the outpatient clinics of
18 UK hospital centres20. It may simply reflect the
fact, demonstrated in previous studies, that the
response of FEV1 measured as a percent of the
baseline value has the worst likelihood ratio to differentiate between responders and not responders
because it over scores bronchodilator response in
patients with low FEV121, 22.
Bronchodilator responsiveness according to
GOLD stage in our study showed that the magnitude of FEV1 response decreased progressively with
increasing disease severity, whereas the magnitude
of the volume response increased as the GOLD
stage was higher. Similar findings we described
by Taskin et al that showed that the percentage
of COPD patients exhibiting a flow response decreased progressively with the increase of GOLD
stage13. At the same time, the association between
flow and volume responses changes as COPD becomes more severe, shown by the significant correlation between the increase of FEV1 and FVC in
milder COPD stages that is lost in the most severe
COPD patients. In a similar way, Schermer et al
have demonstrated that the GOLD stage produces
a ‘shift’ in the association between flow and volume response producing that from GOLD stage I
to GOLD stage III the slope of the regression line
became gradually steeper, which indicated that
a particular FEV1 response was accompanied by
a higher FVC response as the GOLD stage was
more severe23.That decrease of the flow response
and increase of the volume response as the airway
obstruction is more severe may be explained by the
fact that the FEV1 is determined by the airflow
at high to medium lung volumes, whereas FVC is
mainly determined by airway narrowing and flow
limitation at low lung volumes23 .It could mean that
a post bronchodilator change in FVC without a
concomitant change of FEV1 were determined by
the fact that the airway smooth muscle tone—the
site of action of all inhaled bronchodilators—is a
major determinant of airway calibre at low, but
not at high lung volumes8. On the other hand,
Newtonet al23 reported significant increases in
FVC following the administration of salbutamol
in severely and moderately hyperinflated COPD
patients, as the same time that the inspiratory
capacity improved and the functional residual
capacity and residual volume decreased, despite
significant improvements in FEV1 in only a minority of patients.
Cerveri et al, had shown in an elegant study in
10 patients, that there was an inverse relationship
between salbutamol-induced increments in FEV1
and various indexes of emphysema, in spite of conspicuous increments of FVC. They showed that the
airway caliber increased with lung volume in all
FVC-FEV1responders but decreased or remained
unchanged in most FVC responders8. They measured the small-airway caliber by high-resolution
computed tomography at functional residual
capacity and total lung capacity in five chronic
obstructive pulmonary disease patients with an
isolated increase of FVC (FVC responders) and
five with an increase of both FVC and FEV1(FVC-FEV1 responders) after inhalation of salbutamol.
In FVC-FEV1 responders, the airway diameter
increased with the cube root of increase in lung
volume but was unchanged or even decreased in
four of five FVC responders. They concluded that isolated volume response to bronchodilators is a
characteristic of severe emphysema involving more
than 40% of lung parenchyma.
A limitation of our study comes from the design
that was based on the retrospectively reported
standardised data collected in an unselected group
of patients with a clinical and physiological diagnosis of COPD. One of the theoretical limitations
of the studies performed byusing lung function
data obtained for routine diagnostic purposes
in primary care, is the lower proportion of mild
COPD patients. However, more than a quarter of
our patients were classified as GOLD stage I, we
used the old GOLD classification because in the
pulmonary laboratory all the clinical data was not
available in order to classify the patients beyond
the FEV1.
We used only salbutamol (as it is the standard
practice in our laboratory) rather than a combination of salbutamol and ipratropium, which
may have added some additional effects at both
low and high dose combinations25. However, the
additive effect of combining drugs is considered
quite modest and should not have a significant
impact on the results.
We only included patients diagnosed as COPD
by a certified pulmonologist, however, even when
they had data regarding respiratory symptoms,
(self-reported) bronchial hyper responsiveness and
allergies, it is known that none of these characteristics is specific for asthma25 and we cannot exclude that COPD and asthma could have co-existed
within the same patient in some of our subjects.
This may have influenced the rate of bronchial
responsiveness. The exclusion of patients without
a smoking history decreases but not completely
excludes the possibility of some overlap categories
amongst our patients27. An additional limitation
in our study was that we did not include (because
the small proportion of patients in whom it was
available) lung volume and DLCO measurement
to certify the presence of hyperinflation and or
parameters suggestive of emphysema.
This dissociated response in volume and flow,
may explain a frequent clinical finding: that
patients with COPD –even if nonresponders in
terms of FEV1– may benefit from bronchodilators.
It could be the result of being able to breathe at
a lower lung volume due to reduced airtrapping,
although they are still flow limited28. The clinical
significance of these changes in in accordance with the observation that improvements in exercise
endurance and dyspnoea during exercise following
bronchodilator therapy, correlate better with increases in inspiratory capacity than with increases
in FEV129.
In summary, we have identified a significant
number of COPD patients who exhibited an increase only in forced vital capacity after the administration of salbutamol. We have also shown in a
primary care COPD population, that patients with
milder GOLD severity differ from the more severe
in terms of flow (FEV1) and volume (FVC) responses after bronchodilators. Future studies should
identify the reproducibility and most accurate
cut-off values of these volume-based definitions
that appear to be useful in defining the effect of
bronchodilators in symptomatic COPD patients.
1. Pellegrino R, Viegi G, Brusasco V, Crapo RO, Burgos F, Casaburi R, et al. Interpretative strategies for lung function tests. EurRespir J 2005; 26 (5): 948-968.
2. Garay SM. Pulmonary Function Testing. In: Rom WM, editor. Environmental and Occupational Medicine. Philadelphia, Lippincott Williams & Wilkins, 4th edition, 2007. p. 200-235.
3. Ayres JM, Griesbach SJ, Reimold F, Evans RG. (1974) Bronchial component in chronic obstructive pulmonary disease. Am J Med 57: 183-91.
4. Girard WM, Light RW. (1983) Should the FVC be considered in evaluating response to bronchodilators? Chest 84: 87-89.
5. Ramsdell JW, Tisi GM. (1979) Determination of bronchodilatation in the clinical pulmonary function laboratory: role of changes in static lung volumes. Chest 76: 622-628.
6. Quanjer PH, Tammeling GJ, Cotes JE, Pedersen OF, Peslin R, Yernault J-C. (1993) Lung volumes and forced ventilatory flows. EurRespir J 6 Suppl 16: 5-0.
7. Tantucci C, Duguet A, Similowski T, Zelter M, Derenne JP, Milic-Emili J. Effect of salbutamol on dynamic hyperinflation in chronic obstructive pulmonary disease patients. EurRespir J 1998; 12(4): 799-8047.
8. Cerveri1, Pellegrino R, Dore R, Corsico A, Fulgoni P, van de Woestijne KP, BrusascoV. Mechanisms for isolated volume response to a bronchodilator in patients with COPD. J Appl Physiol (1985).2000 Jun; 88(6): 1989-1995.
9. M.R.Miller, J.Honkinson, V.Brusasco, F.Burgos, R.Casaburi, A. Coates, R. Standarisation of spirometry. EurRespir J 2005; 26: 319-338.
10. American Thoracic Society (1991) Lung function testing: selection of reference values and interpretative strategies. Am Rev Respir Dis 144: 1202-1218.
11. Tashkin D, Kesten S. Long-term treatment benefits with tiotropium in COPD patients with and without short-term bronchodilator responses. Chest 2003; 123: 1441-1449.
12. Global Initiative for Chronic Obstructive Lung Disease (GOLD) en http//www.goldcopd.com (accessed: 30.1.06).
13. Tashkin DP1, Celli B, Decramer M, Liu D, Burkhart D, Cassino C, Kesten S. Bronchodilator responsiveness in patients with COPD. EurRespir J. 2008 Apr; 31(4): 742-750.
14. Brand PL, Quanjer PH, Postma DS, et al. Interpretation of bronchodilator response in patients with obstructive airways disease. The Dutch Chronic Non-Specific Lung Disease (CNSLD) Study Group. Thorax 1992; 47: 429-436.
15. Dompeling E, Van Schayck CP, Molema J, et al. A compari- son of six different ways of expressing the bronchodilating response in asthma and COPD: reproducibility and dependence of prebronchodilator FEV1. EurRespir J 1992; 5: 975-981
16. Quadrelli S, Roncoroni AJ, Montiel GC Response to bronchodilators among patients with bronchial asthma]. Medicina (B Aires). 1994; 54(4): 311-8.
17. Burge PS, Calverley PM, Jones PW, et al. Randomised, double blind, placebo controlled study of fluticasone propionate in patients with moderate to severe chronic obstructive pulmonary disease: the ISOLDE trial. BMJ 2000; 320: 1297-303.
18. Lofdahl CG, Postma DS, Laitinen LA, et al. The European Respiratory Society study on chronic obstructive pulmonary disease (EUROSCOP): recruitment methods and strategies. Respir Med 1998; 92: 467-472.
19. Walker PP, Calverley PM. The volumetric response to bronchodilators in stable chronic obstructive pulmonary disease. COPD. 2008 Jun; 5(3): 147-152.
20. Calverley PM, Burge PS, Spencer S, Anderson JA, Jones PW. Bronchodilator reversibility testing in chronic obstructive pulmonary disease. Thorax. 2003 Aug; 58(8): 659-664.
21. Quadrelli SA, Roncoroni AJ, Montiel GC. Evaluation of bronchodilator response in patients with airway obstruction. RespirMed. 1999 Sep; 93(9): 630-6.
22. Dompeling E, Van Schayck CP, Molema J, et al. A comparison of six different ways of expressing the bronchodilating response in asthma and COPD: reproducibility and dependence of prebronchodilator FEV1. EurRespir J 1992; 5: 975-981.
23. Schermer T, Heijdra Y, Zadel S, van den Bemt L, Boonmande Winter L, Dekhuijzen R, SmeeleI. Flow and volume responses after routine salbutamol reversibility testing in mild to very severe COPD. Respir Med. 2007 Jun; 101(6): 1355-1362.
24. Newton MF, O’Donnell DE, Forkert L. Response of lung volumes to inhaled salbutamol in a large population of patients with severe hyperinflation. Chest 2002; 121: 1042-1050].
25. Hadcroft J, Calverley PMA. Alternative methods for assessing bronchodilator reversibility in chronic obstructive pulmonary disease. Thorax 2001; 56: 713-720.
26. Chanez P, Vignola AM, Shaugnessy T, Enander I, Li D, Jeffery PK, et al. Corticosteroid reversibility in COPD is related to features of asthma. Am J RespirCrit Care Med 1997; 155: 1529-1534.
27. Grootendorst DC, Rabe KF. Mechanisms of bronchial hyperreactivity in asthma and chronic obstructive pulmonary disease. Proc Am ThoracSoc 2004; 1: 77-87.
28. Tantucci C, Duguet A, Similowski T, Zelter M, Derenne JP, Milic-Emili J. Effect of salbutamol on dynamic hyperinflation in chronic obstructive pulmonary disease patients. EurRespir J 1998; 12(4): 799-8047.
29. O’Donnell D, Fluge T, Gerken F, et al. Effects of tiotropium on lung hyperinflation, dyspnoea and exercise tolerance in COPD. EurRespir J 2004; 23: 832-840.